Le Infezioni in Medicina, n. 2, 233-238, 2025
doi: 10.53854/liim-3302-10
CASE REPORTS
Strongyloides stercoralis hyperinfection with shock and thrombosis
Darío Sebastián López-Delgado1, Carlos A. Narváez2, Roberto Bonfante-Villalobos3, Joshua Emmanuel Arteaga3, Hector Fabio Sanchez-Galvez4, Yamile Jurado4, Alfonso J. Rodriguez-Morales5,6
1Grupo Interdisciplinario de Investigación Salud Enfermedad (GIISE), Universidad Cooperativa de Colombia. San Juan de Pasto, Nariño, Colombia;
2Universidad Nacional Autónoma de Nicaragua, Managua, Nicaragua;
3Universidad Cooperativa de Colombia, San Juan de Pasto, Nariño, Colombia;
4Hospital Universitario Departamental de Nariño, Pasto, Nariño, Colombia;
5Faculty of Health Sciences, Universidad Científica del Sur, Lima 15067, Peru;
6Grupo de Investigación Biomedicina, Faculty of Medicine, Fundación Universitaria Autónoma de las Américas-Institución Universitaria Visión de las Américas, Pereira Risaralda 660003, Colombia.
Article received 24 March 2025 and accepted 5 May 2025
Corresponding author
Alfonso J. Rodriguez-Morales
E-mail: arodriguezmo@cientifica.edu.pe
SummaRY
Strongyloides stercoralis (SS) is an intestinal parasite that can cause chronic asymptomatic infections, but in rare cases, it can progress to hyperinfection syndrome (SHS). This report describes a case of SHS associated with deep vein thrombosis and pulmonary thromboembolism, a rare manifestation in an immunocompetent patient. A 19-year-old female patient with a 15-day history of abdominal pain, progressive edema of the lower limbs, hemoptotic cough, asthenia, and weight loss. During her hospitalization, she developed sudden dyspnea, desaturation, and distributive shock, requiring invasive mechanical ventilation. Pulmonary angiotomography showed pulmonary thromboembolism, and deep vein thrombosis was diagnosed. Bronchoscopy revealed alveolar hemorrhage, while bronchoalveolar and duodenal lavage confirmed the presence of SS. Ivermectin and albendazole were started with full-dose anticoagulation for the thrombotic event. After one week of management, bronchoalveolar lavage results were negative, and the patient showed significant improvement with no long-term complications. SHS is rare in immunocompetent patients, and its association with thrombosis has been poorly documented in the literature. This case emphasizes the importance of early diagnosis and timely management to avoid life-threatening complications. It also highlights the need for surveillance in endemic regions and the appropriate use of evidence-based therapeutic strategies.
Keywords: strongyloidiasis, parasitic diseases, opportunistic infections, venous thromboembolism, anthelmintics.
INTRODUCTION
Strongyloides stercoralis (SS), commonly known as threadworm, is a soil-transmitted nematode belonging to the roundworm group [1]. The prevalence of SS varies by geographic region, exceeding 50% in tropical areas and ranging from 10% to 15% in other countries. Additionally, some studies have reported a slightly higher prevalence in women compared to men [2].
One of the rare complications of infection by the parasite Strongyloides stercoralis is Hyperinfection Syndrome (SHS). This syndrome causes an imbalance in the host immune response and can lead to severe systemic complications, such as multiorgan failure, sepsis, or thrombosis [3]. This complication was first described in 1966, with a post-mortem diagnosis [4].
This article describes a case of SHS in an immunocompetent patient, with its rare association with deep vein thrombosis and pulmonary thromboembolism. Diagnostic and therapeutic challenges are discussed and contextualized in the literature to enhance the understanding of SHS in patients without immunosuppression and to emphasize the importance of timely diagnosis and treatment in endemic regions.
CASE REPORT
A 19-year-old female patient was admitted to a high-complexity hospital in Nariño, Colombia, after being referred for a 15-day history of abdominal pain located in the mesogastrium and hypogastrium. Her symptoms were accompanied by progressive lower limb edema, nausea, vomiting, hemoptoic cough, asthenia, adynamia, and an unquantified fever. She had also experienced an approximate weight loss of 2 kilograms over two weeks.
On admission, physical examination revealed a patient in fair general condition with adequate nutritional status. Vital signs showed hypotension (blood pressure 79/43 mmHg), tachycardia (110 beats per minute), respiratory rate of 20 breaths per minute, and normothermia (36.2°C). She weighed 50 kg, was 1.53 m tall, and had a body mass index (BMI) of 21 kg/m². A head-to-toe examination revealed grade II bimalleolar edema, splenomegaly, and a gallop rhythm on cardiac auscultation.
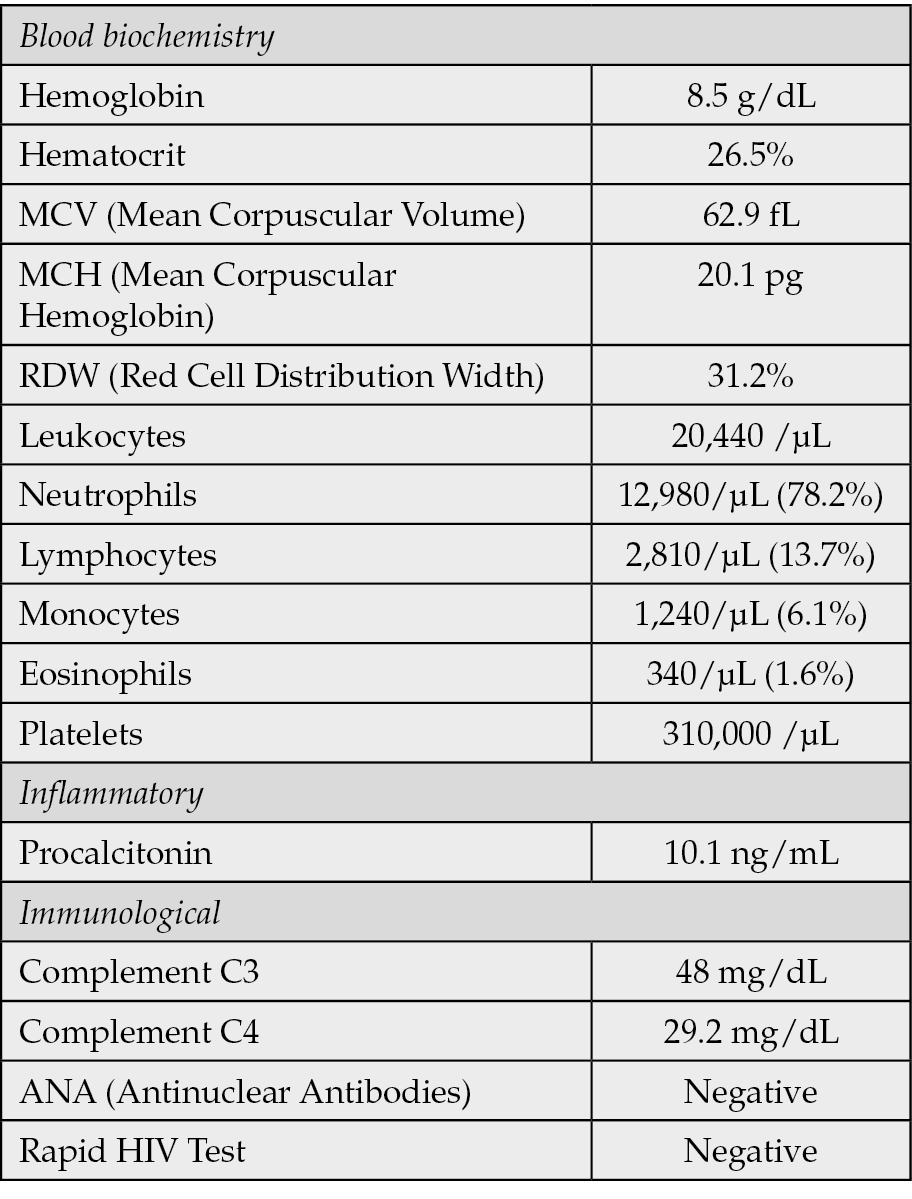
At the referring center, a myeloproliferative syndrome had initially been suspected based on an abdominal CT scan that showed mesenteric lymphadenopathy with reactive features and hypochromic microcytic anemia. Initial laboratory results (Table 1) revealed leukocytosis with a left shift, elevated procalcitonin levels, and features consistent with a systemic inflammatory response. Empirical antibiotic therapy was started, but the patient remained hypotensive despite intravenous fluid resuscitation, requiring vasopressor support and subsequent admission to the intensive care unit due to distributive shock.
Table 1 - Laboratory Findings.
During hospitalization, the patient developed sudden dyspnea and oxygen desaturation, prompting the initiation of invasive mechanical ventilation. A pulmonary angiotomography (Figures 1a and 1b) revealed pulmonary interstitial infiltrates, pulmonary thromboembolism, and deep vein thrombosis. Full-dose anticoagulation therapy was started accordingly.
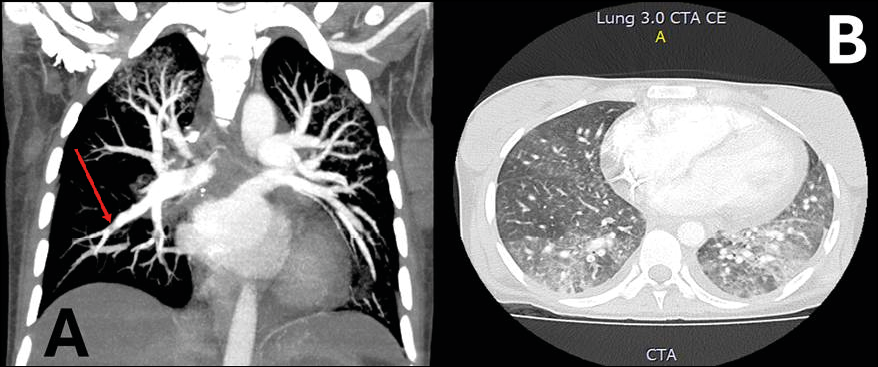
Figure 1 - Pulmonary Angiotomography: Coronal (A) and Axial (B) images. A. Central filling defect in the branches of the basal segment of the right lower lobe, with signs of a “lane” indicative of pulmonary thromboembolism. B. Interstitial involvement in medial areas with a ground-glass appearance.
In light of the thrombotic episode in a patient without identifiable risk factors, an immunological workup was performed, which showed no significant abnormalities. A bronchoscopy was performed because of the pulmonary findings, revealing diffuse alveolar hemorrhage. Concurrently, abdominal CT findings suggestive of intestinal inflammation prompted an upper gastrointestinal endoscopy, which showed extensive gastric and duodenal mucosa ulcerations.
Two days after obtaining samples via bronchoalveolar lavage (BAL) and duodenal biopsy, pathology confirmed the presence of helminths in both specimens (Figures 2a and 2b), with morphological features compatible with SS.
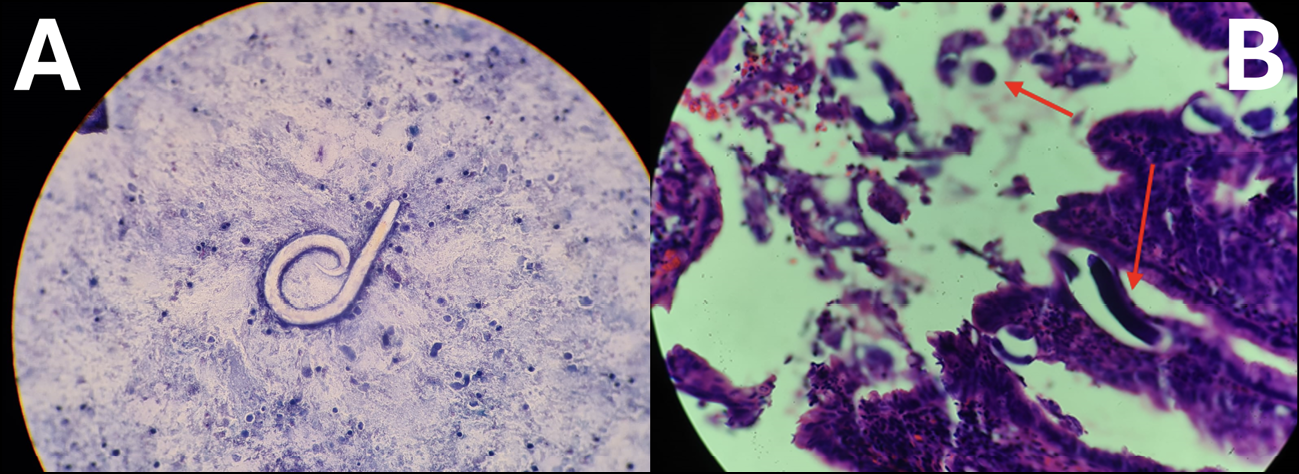
Figure 2 - Strongyloides stercoralis in bronchoalveolar lavage (A) and duodenum (B). The images show structures compatible with the parasite in bronchoalveolar lavage (A) and duodenal mucosa (indicated by red arrows) (B). Image A: saline wet mount, 400x magnification. Image B: H&E stain, 400x magnification.
A combined antiparasitic regimen was initiated with ivermectin at 200 µg/kg/day (via nasogastric tube) and albendazole 400 mg twice daily. Anticoagulation was continued. Mechanical ventilation was successfully discontinued within 2 to 3 days of starting treatment, and vasopressor support was gradually withdrawn.
After two weeks of therapy, the patient had significantly improved and could resume oral intake and medications. A follow-up bronchoscopy revealed no evidence of larval persistence. Although serology studies and agar culture were not performed due to resource limitations in our center, the clinical and parasitological response was favorable. The patient was discharged during the third week of hospitalization, and antiparasitic treatment was stopped. Anticoagulation therapy was continued for three months following discharge.
The patient showed no signs of relapse or complications at the six-month follow-up. HTLV-1 serology was negative, and imaging confirmed complete recanalization of the previously affected deep venous system.
DISCUSSION
SHS is a severe and potentially fatal complication of infection with this parasitic nematode. It is characterized by uncontrolled proliferation in the intestine and massive migration to other organs, such as the lungs, liver, and central nervous system [3].
Risk factors commonly described in the literature, such as the use of corticosteroids or infection by HTLV-1, were not identified in this patient [5, 6]. The latter appears to be associated with alterations in regulatory T-cell counts and reduced antigen-induced IL-5 production, resulting in decreased levels of total IgE [5]. A systematic review and meta-analysis found a strong association between the HTLV-1 virus and SHS, with an odds ratio (OR) of 59.9 (95% CI: 18.1-198). A higher treatment failure rate was also observed, with an OR of 5.05 (95% CI: 2.50-10.17).
Given the above, screening for HTLV-1 was performed in this patient, with negative results. In addition, other risk factors associated with SHS have been identified, including solid organ transplantation and hematopoietic transplantation, as well as alcoholism, which may predispose to SHS through increased endogenous cortisol levels, reduced intestinal motility and protein-calorie malnutrition, which compromises both innate and acquired immunity, decreasing the Th2 lymphocyte response [8-10]. Likewise, the presence of SHS has been described in individuals with apparently normal anthropometric parameters but with severe vitamin deficiencies [11]. An association between the development of SHS and the use of immunosuppressive drugs, such as azathioprine, cyclophosphamide, methotrexate, tacrolimus, bleomycin, carmustine, chlorambucil, doxorubicin, daunorubicin, ifosfamide, melphalan, and mitoxantrone, has also been documented [3, 12]. Despite these risk factors, the clinical case showed no evidence of immunological alterations or immunosuppression.
Eosinophilia is a common finding in SS infection, but not all patients with SS infection present with eosinophilia. Although eosinophilia is reported in 55% to 77% of cases at diagnosis, its absence does not rule out infection, particularly in immunocompromised individuals or those in endemic areas [13, 14]. In such settings, repeated exposure or altered immune responses may lead to normal eosinophil counts despite active infection, making eosinophilia an unreliable screening tool [15]. Clinical suspicion and specific diagnostic testing remain essential.
Although SHS is more frequently reported in immunosuppressed patients, it can also affect immunocompetent individuals, particularly those living in endemic regions. This emphasizes the importance of maintaining a high index of suspicion, especially in patients presenting with atypical symptoms or unexplained thrombotic events. From a public health standpoint, these cases highlight the need for increased awareness, improved surveillance, and targeted screening strategies in tropical areas where Strongyloides is endemic.
The clinical manifestations of SHS can be diverse, affecting various organs and systems, including the gastrointestinal, cutaneous, cardiovascular, renal, central nervous, and pulmonary systems [3, 16, 17]. However, one of the most infrequent manifestations is thrombosis. Eleven cases of thrombotic phenomena associated with hyperinfection syndrome are reported in the current literature, including post-necropsy pulmonary thrombosis, portal and biliary thrombosis, intraventricular thrombus, pulmonary and renal artery thrombosis, lower limb and abdominal aorta thrombosis, cardiac thrombosis, and mesenteric thrombosis [1, 3, 4, 16, 18-22]. In this case, thrombosis presented as extensive involvement of the iliac and femoral veins and pulmonary thromboembolism. While thrombotic events have been previously reported in association with SHS, they are uncommon, particularly in young, immunocompetent individuals. The concurrent presence of alveolar hemorrhage further highlights the diverse and severe spectrum of SHS manifestations. This case contributes to the growing body of evidence and reinforces the need to consider such complications in endemic areas, even in patients without classic risk factors. The hypercoagulability observed may be linked to cytokine-mediated inflammation and eosinophilic endothelial infiltration [11, 18, 20].
Concomitantly, the patient developed a clinical presentation of alveolar hemorrhage, which led to the performance of a BAL, where the presence of the parasite was evidenced. This manifestation is more frequent compared to the thrombotic presentation. Some local reports describe a similar pathophysiological pathway, in which it has been theorized that the passage and infiltration of parasites into the alveoli cause hemorrhage, which subsequently leads to ventilatory failure in the patient [23].
In this case, stool parasitology was not performed due to SHS’s initially low clinical suspicion. The diagnosis was ultimately established based on detecting SS larvae in bronchoalveolar lavage fluid and was further supported by histopathological findings in intestinal biopsies. Nevertheless, identifying larvae in stool is considered the gold standard for confirming infection, especially in cases of hyperinfection, where timely and accurate diagnosis is vital [5]. However, conventional stool microscopy has limited sensitivity, often missing up to 70% of cases with a single sample, particularly in chronic or low-burden infections. Sensitivity can be markedly improved by analyzing multiple stool samples and using enhanced techniques such as agar plate culture or the Baermann method [24].
Direct visualization remains the most effective method for diagnosis. Unlike other intestinal parasites, SS is characterized by a low parasite load and intermittent larval shedding [25, 26]. Therefore, current diagnostic strategies include:
1) direct visualization of larvae by biological fluid microscopy,
2) identification of larvae in tissue biopsies,
3) serological tests,
4) molecular assays [1].
Regarding treatment, the Public Health Agency of Canada’s Tropical Medicine and Travel Advisory Committee (CATMAT) established in 2016 the following recommendations: ivermectin 200 µg/kg/day, orally or subcutaneously, once daily, plus albendazole 400 mg twice daily, until cessation of larval shedding and clinical improvement [27]. These recommendations were based on WHO management guidelines for strongyloidiasis (https://www.who.int/teams/control-of-neglected-tropical-diseases/soil-transmitted-helminthiases/strongyloidiasis) and the effectiveness observed in SHS case reports and case series. In this case, one week after initiation of treatment, the patient showed marked improvement. Bronchial lavage performed on the seventh day was negative for the parasite, and medical discharge was subsequently granted.
SHS is a life-threatening condition, with mortality rates as high as 60%, according to some authors, and as high as 80-100% in the absence of efficacious treatment [17, 28]. In a study that included a systematic review of 244 case reports, a mortality rate of 60% was recorded. Untreated patients had up to 100% mortality, while those treated with albendazole, thiabendazole, and ivermectin had lower mortality rates of 73%, 51%, and 47%, respectively [29].
CONCLUSIONS
SHS is a serious condition with a rare association with thrombosis, especially in immunocompetent patients. This case highlights the importance of early diagnosis in endemic regions and its inclusion in the differential diagnosis of unexplained thrombotic events. Treatment with ivermectin and albendazole was effective, allowing long-term, uncomplicated recovery. However, further studies are needed to investigate the relationship between SHS and hypercoagulability. This report reinforces the need for improved management strategies and medical guidelines. Documenting atypical cases is key to advancing knowledge of the disease.
Author contributions
dSL: Conceptualization, Project Management, Visualization, Writing - original draft; CAN: Conceptualization, Supervision, Visualization, Writing - revision and editing; RBV: Conceptualization, Visualization, Writing - original draft; JEA: Conceptualization, Visualization, Writing - original draft; HFSG: Conceptualization, Visualization, Writing - original draft; YJ: Conceptualization, Supervision, Writing - proofreading and editing; AJRM: Conceptualization, Supervision, Writing - proofreading and editing.
Conflicts of interest
The authors declare that they have no conflict of interest.
Funding
The authors did not receive any specific grant from public, commercial, or not-for-profit funding agencies.
Ethics approval and informed consent
the research group of the Hospital Universitario Departmental de Nariño approved this work. The patient gave informed consent for publication. The research regulations of the Nuremberg Codes, the Helsinki Declaration, the Belmont Report, and resolution 8430 of 1993 for research in Colombia were followed.
Acknowledgements
The authors acknowledge the collaboration between the patient, his family, and the Hospital Universitario Departmental de Nariño work team. Dr Rodriguez-Morales would like to dedicate this publication to the memory of Prof. Olinda Delgado, PhD (1930-2024), an expert in tropical and parasitic diseases such as leishmaniasis and toxocariasis, from Venezuela, her major mentor and excellent human being, who passed away in Caracas, December 2024, R.I.P. This article has been registered in the Research Proposal Registration of the Coordination of Scientific Integrity and Surveillance of Universidad Cientifica del Sur, Lima, Peru, under the number PI-50-2025-0268.
REFERENCES
[1] Mora CS, Segami MI, Hidalgo JA. Strongyloides stercoralis hyperinfection in systemic lupus erythematosus and the antiphospholipid syndrome. Semin Arthritis Rheum. 2006; 36(3): 135-143.
[2] Schär F, Trostdorf U, Giardina F, et al. Strongyloides stercoralis, Global Distribution and Risk Factors. PLoS Negl Trop Dis. 2013; 7(7): e2288.
[3] Karanam LS, Basavraj GK, Papireddy CKR. Strongyloides stercoralis hyperinfection syndrome. Indian J Surg. 2021; 83: 582-586.
[4] Cruz T, Reboucas G, Rocha H. Fatal strongyloidiasis in patients receiving corticosteroids. N Engl J Med. 1966; 275(20): 1093-1096.
[5] Krolewiecki A, Nutman TB. Strongyloidiasis. Infect Dis Clin North Am. 2019; 33(1): 135-151.
[6] Vadlamudi RS, Chi DS, Krishnaswamy G. Intestinal strongyloidiasis and hyperinfection syndrome. Clin Mol Allergy. 2006; 4: 8.
[7] Ye L, Taylor GP, Rosadas C. Human T-cell lymphotropic virus type 1 and Strongyloides stercoralis co-infection, a systematic review and meta-analysis. Front Med (Lausanne). 2022; 9: 832430.
[8] Kim JH, Kim DS, Yoon YK, Sohn JW, Kim MJ. Donor-derived Strongyloidiasis infection in solid organ transplant recipients, a review and pooled analysis. Transplant Proc. 2016; 48(7): 2442-1449.
[9] Abdalhamid BA, Al Abadi AN, Al Saghier MI, Joudeh AA, Shorman MA, Amr SS. Strongyloides stercoralis infection in kidney transplant recipients. Saudi J Kidney Dis Transpl. 2015; 26(1): 98-102.
[10] Takele Y, Adem E, Getahun M, et al. Malnutrition in healthy individuals results in increased mixed cytokine profiles, altered neutrophil subsets and function. PLoS One. 2016; 11(8): e0157919.
[11] Marathe A, Date V. Strongyloides stercoralis hyperinfection in an immunocompetent patient with extreme eosinophilia. J Parasitol. 2008; 94(3): 759-60.
[12] Yeung S, Bharwada Y, Bhasker S, Boggild A. Strongyloidiasis, what every gastroenterologist needs to know. Ther Adv Chronic Dis. 2022; 13, 204062232211374.
[13] Buonfrate D, Fittiplado A, Vlieghe E, Bottieau E. Clinical and laboratory features of Strongyloides stercoralis infection at diagnosis and after treatment, a systematic review and meta-analysis. Clin Microbiol Infect. 2021; 27(11): 1621-1628.
[14] González A, Gallo M, Valls M, et al. Clinical and epidemiological features of 33 imported Strongyloides stercoralis infections. Trans R Soc Trop Med Hyg. 2010; 104(9): 613-616.
[15] Hays R, Thompson F, Esterman A, McDermott R. Strongyloides stercoralis, eosinophilia, and type 2 diabetes mellitus, the predictive value of eosinophilia in the diagnosis of S. stercoralis infection in an endemic community. Open Forum Infect Dis. 2016; 3(1): ofw029.
[16] Thaden J, Cassarino DS, Truong Q, Dickson B. Eosinophilic endocarditis and Strongyloides stercoralis. Am J Cardiol. 2013; 112(3): 461-462.
[17] Vasquez-Rios G, Pineda-Reyes R, Ruiz EF, Terashima A. Strongyloides stercoralis hyperinfection syndrome, a deeper understanding of a neglected disease. J Parasit Dis. 2019; 43(2): 167-175.
[18] Filkins LM, Choksi TT, Patel D, Nowalk AJ. Biliary Strongyloides stercoralis with cholecystitis and extensive portal vein thrombosis. Open Forum Infect Dis. 2017; 4(4): ofx217.
[19] Vlug M. Hepatocellular carcinoma and Strongyloides infection with portal vein thrombosis. J Liver Res Disord Ther. 2018; 4(1): 13-14.
[20] Al Hadidi M, Shaaban H, Jumean KH, Peralta P. Loeffler’s syndrome secondary to hyperinfection by Strongyloides stercoralis associated with methotrexate in a patient with rheumatoid arthritis. J Glob Infect Dis. 2018; 10(1): 29-30.
[21] Waters M, Krajden S, Rivest C, Tsega Y. Case report, two cases of strongyloidiasis presenting with thrombotic events. Am J Trop Med Hyg. 2019; 101(2): 418-421.
[22] Stam-Slob MC, Cornelissen MM, Scheper H, Nellen JF, Visser LG. Thrombotic micro-angiopathy due to Strongyloides stercoralis hyperinfection. Clin Infect Pract. 2022; 15: 100153.
[23] Rodríguez-Pérez EG, Arce-Mendoza AY, Saldívar-Palacios R, Escandón-Vargas K. Fatal Strongyloides stercoralis hyperinfection syndrome in an alcoholic diabetic patient from México. Biomédica. 2020; 40(1): 32-36.
[24] Luvira V, Watthanakulpanich D, Pittisuttithum P. Management of Strongyloides stercoralis, a puzzling parasite. Int Health. 2014; 6(4): 273-281.
[25] Barros N, Montes M. Infection and hyperinfection with Strongyloides stercoralis, clinical presentation, etiology of disease, and treatment options. Curr Trop Med Rep. 2014; 1: 223-228.
[26] Topić MB, Grubišić B, Kovačević E, Sviben M, Santini M. Strongyloidiasis – diagnostic and therapeutic dilemmas in hyperinfection patients, a case series. J Helminthol. 2022; 96: e76.
[27] Boggild AK, Libman M, Greenaway C, McCarthy AE; Committee to Advise on Tropical Medicine and Travel (CATMAT). CATMAT statement on disseminated strongyloidiasis, prevention, assessment and management guidelines. Can Commun Dis Rep. 2016; 42(1): 12-19.
[28] Henriquez-Camacho C, Gotuzzo E, Echevarria J, et al. Ivermectin versus albendazole or thiabendazole for Strongyloides stercoralis infection. Cochrane Database Syst Rev. 2016; 2016(1): CD007745.
[29] Buonfrate D, Requena-Mendez A, Angheben A, et al. Severe strongyloidiasis, a systematic review of case reports. BMC Infect Dis. 2013; 13: 78.