Le Infezioni in Medicina, n. 1, 29-49, 2025
doi: 10.53854/liim-3301-4
REVIEWS
Updates on HIV Pre-exposure Prophylaxis in Latin America: Available Drugs and Implementation Status
Christian Gonzales-Gavancho1, Julieta M. Araoz-Salinas1,2, Ruth Ramon Tapia3, Carlos Quispe-Vicuña4, Martin E. Reategui-Garcia5, Wagner Rios-Garcia6, Maria Jose Baltodano-Calle7, Ivana Belanovic-Ramirez1,8, Allison N. Ortiz-Pardo1, Brando Ortiz-Saavedra9, Victor Y. M. Campos1,10, Jorge Alave11, Jose A. Gonzales-Zamora3
1Peruvian American Medical Society (PAMS), Albuquerque, NM 87111, USA;
2Asociacion Civil Impacta Salud y Educación, Lima 15063, Perú;
3Division of Infectious Diseases, Department of Medicine, Miller School of Medicine, University of Miami, Miami, FL 33136, USA;
4Grupo de Investigación Neurociencias, Metabolismo, Efectividad Clínica y Sanitaria (NEMECS), Universidad Científica del Sur, Lima 150142, Perú;
5Universidad Nacional de la Amazonia Peruana, Iquitos 16000, Perú;
6Universidad Nacional San Luis Gonzaga, Ica 11001, Perú;
7Universidad Peruana Cayetano Heredia, Lima 15012, Perú;
8Universidad Peruana de Ciencias Aplicadas, Lima 15023 Perú;
9Universidad Nacional de San Agustín de Arequipa, Arequipa 040101, Perú;
10Department of Community Health and Family Medicine. University of Florida, College of Medicine, Jacksonville, Florida 32209, USA;
11Escuela de Medicina, Universidad Peruana Union, Lima 15464, Perú.
Article received 30 October 2024 and accepted 28 January 2025
Corresponding author
Jose A. Gonzales-Zamora
E-mail: jxg1416@med.miami.edu
SummaRY
Latin America has reported a 9% increase in new HIV infections from 2010 to 2023. Pre-exposure prophylaxis (PrEP) is a crucial biomedical intervention for preventing HIV transmission. Currently, several antiretroviral drugs, in various forms of administration, have demonstrated high efficacy and effectiveness to protect against HIV. Among the oral drugs, we have emtricitabine/tenofovir and emtricitabine/tenofovir alafenamide, while alternative options include the dapivirine vaginal ring, injectable drugs such as Cabotegravir, and the most recently studied Lenacapavir. Despite their high efficacy and effectiveness, implementing PrEP in Latin America has been challenging throughout the region. Although some countries such as Brazil, Mexico, and Colombia have shown progress in increasing the number of users, there is a significant gap between these countries and others where PrEP access remains limited or non-existent. Barriers such as lack of awareness, inadequate funding, political instability, and outdated policies contribute to disparities in access, leaving many populations at high risk of HIV infection without this preventative measure. Innovative strategies need to be implemented to address and monitor policies that ensure access for all at-risk populations.
Keywords: HIV prevention, pre-exposure prophylaxis, implementation, Latin America.
INTRODUCTION
According to the World Health Organization (WHO), HIV infections in 2023 have been reduced by 60% since their peak in 1995; however, approximately 39.9 million people were still living with HIV by the end of 2023 [1]. Globally, the WHO and Joint United Nations Programme on HIV/AIDS (UNAIDS) aim for 95% of people living with HIV to know about their HIV status, to receive antiretroviral treatment (ART), and to achieve viral suppression. This is complemented by reducing new HIV infections to fewer than 370,000 per year and AIDS-related deaths to fewer than 250,000 per year by 2025 [2, 3].
Effective control of this infection requires medical interventions and fundamental social and political changes [4]. In this respect, the UNAIDS strategy (2021-2026) highlights the need to improve the implementation and funding of HIV preventive measures [5]. These targets point to the need for an integrated approach that addresses health inequalities, improves access to care, and strengthens healthcare systems. Considerable improvement has been achieved globally, with a decrease of 51% in the annual number of AIDS-related deaths between 2010 and 2023. However, some regions fall short, such as Latin America (LA) with only a 28% decrease [6]. Additionally, in 2022 the Pan American Health Organization (PAHO) reported that only around 72% of people with HIV were receiving ART in LA. This represented a notable increase compared to the 56% seen in 2016, but it is certainly not enough to reach the global goal [7].
One of the HIV prevention strategies is taking antiretroviral drugs before HIV exposure, which is called Pre-exposure Prophylaxis (PrEP) [8]. This strategy has emerged as a pivotal biomedical intervention in the prevention of HIV transmission. PrEP is being ensured worldwide for its protective effect on the population at high risk of HIV infection, which includes men who have sex with men (MSM), transgender individuals, people with multiple partners or who do not use condoms, sexual workers, people living in locations where the incidence of HIV is 3% or greater, and individuals who have had sexually transmitted infections or who share injection drug needles, amongst others [9].
Despite its high effectiveness, it has been a challenge to implement PrEP adequately and sustainably due to the lack of solid policies that address the current problems such as social stigma and negative connotations to HIV-related treatment and the lack of health environments that guarantee an appropriate supply of these medications for providers and health facilities [10-11]. Also, it is essential to monitor the patient’s adherence to treatment, exposure to HIV, and serological HIV tests to correctly assess the effectiveness of PrEP, all of which are difficult to accomplish in resource-limited settings [12, 13].
Up to 2023, only 67% of countries in LA and the Caribbean had policies approving daily oral PrEP for HIV prevention, which included specific key populations such as MSM, transgender women (TGW), sex workers, and serodiscordant couples [14]. Although most countries in LA have made progress in their fight against HIV, there has been a 9% increase in new HIV infections from 2010 to 2023 [6]. PAHO reported 156,890 people on PrEP during 2023, a three times increase compared to that in 2021, we have to keep in mind that this large number was mainly represented by Brazil with 110,427 people [7]. On the other hand, countries such as Nicaragua and Uruguay have reported only 196 and 240 people on PrEP respectively [7]. Although the overall control of this epidemic seems to be heading in the right direction globally, countries in LA still have a long way to go to implement adequate public health plans for PrEP. Even in the same region, there is a big gap between some countries regarding their policies to prevent and control the spread of HIV. For this reason, the present article summarizes the available evidence for the current drugs and the implementation status of PrEP for HIV in LA.
METHODOLOGY
In the present study, we provide a description of the most representative clinical trials that support the use of antiretroviral drugs for PrEP. We conducted a narrative review of the main aspects of HIV PrEP in LA, including implementation status, cost-effectiveness analysis, and interventions to improve coverage. The search for information was carried out in the Pubmed database and Scopus. In addition, a search of gray literature was conducted through Google to evaluate the clinical practice guidelines from the ministries of health of each country, as well as the official websites of UNAIDS and PAHO.
CURRENT DRUGS FOR HIV PRE-EXPOSURE PROPHYLAXIS
In this section, we will describe the main aspects of current drugs approved for HIV PrEP, focusing on those that are commercially available and supported by efficacy data highlight key findings from Phase 3 clinical trials, as well as ongoing implementation studies that evaluate them in real-world settings. A summary of these drugs and their efficacy data is presented in Table 1.
Table 1 - Characteristics of current HIV PrEP regimens.

Tenofovir disoproxil fumarate-Emtricitabine
Oral Tenofovir disoproxil fumarate-Emtricitabine (TDF-FTC) is an antiretroviral combination of the nucleoside reverse transcriptase inhibitor (NRTI) class used for the treatment and prevention of HIV by blocking viral DNA synthesis and interrupting HIV replication [15]. This drug has been approved for the treatment of HIV by the U.S. Food and Drug Administration (FDA) since August 2004 under the trade name of Truvada® and for PrEP in high-risk individuals in July 2012, becoming the first drug approved for this purpose [16, 17]. According to the latest CDC guidelines released in 2021, oral PrEP with TDF-FTC is recommended for daily use by adolescents and adults at significant risk for HIV infection [18]. On its end, the WHO, since 2015, has recommended the use of TDF/FTC in low- and middle-income countries [19].
There are several clinical trials evaluating the efficacy of this drug depending on the type of population. The PrEP study, a randomized trial with 4747 heterosexual couples, found a 75% reduction in HIV-1 incidence in the group treated with TDF-FTC (95% CI, 55 to 87; P<0.001) [20]. For MSM, The iPrEx study, a randomized trial with 2499 MSM patients, reported a 44% reduction in HIV incidence (95% CI 15-63, p=0.005) [21]. Regarding people who inject drugs, the Bangkok Tenofovir study, a Phase 3 trial with 2413 participants, found a 48.9% reduction in HIV incidence (95% CI: 9.6-72.2; p = 0.01) [22].
In terms of effectiveness, a real-world study conducted in three metropolitan areas of the United States (USA) with a cohort of 557 MSM and TGW patients receiving TDF-FTC showed a mean adherence to PrEP of 82% and the incidence of HIV infection of 0.43 cases per 100 person-years (95% CI: 0.05-1.54). Of the 19 serious adverse events reported, none were related to the use of TDF-FTC [23]. Similarly, the ImPrEP study, a large prospective study with 9509 participants (gay, bisexual, and other cisgender men) from LA (Brazil, Mexico, and Peru), found that very few participants discontinued PrEP (0.91%), with 0.55% discontinuing it due to adverse events [24].
Among the various effects, TDF-FTC use in HIV-uninfected patients was associated with a small but statistically significant decrease in bone mineral density at 24 weeks (-1.42% at the spine and -0.85% at the hip) [25]. Similarly, Among MSM and TGW, a significant increase in mean creatinine by 4.6% and a decrease in creatinine elevation by 3.0% were reported up to 12 weeks [26].
In addition to daily therapy, event-based (on-demand) PrEP dosing is another strategy for preventing HIV. Although not FDA-approved, it is endorsed by the International Antiviral Society of the USA, WHO, and CDC, especially for cisgender MSM patients before practice sex without a condom [27-29]. The use of event-based was evaluated in the IPERGAY study, a randomized trial with 400 MSM who engage in condomless anal sex, found an 86% reduction in HIV transmission (95% CI: 40-98) [30]. In addition, a prospective cohort study of 3065 MSM found no difference in HIV-1 incidence between participants using daily PrEP and those using on-demand PrEP (incidence rate ratio: 1.00, 95% CI: 0.13-7.49; p=0.99) [31].
Tenofovir alafenamide-Emtricitabine
Emtricitabine-tenofovir alafenamide (TAF-FTC) is similar to TDF-FTC but with a more rapid delivery of the active metabolite (tenofovir diphosphate) to peripheral blood mononuclear cells, achieving concentrations at least four times higher, leading to a greater antiviral effect. [32]. TAF-FTC was approved by the U.S. FDA in April 2016 for HIV treatment under the trade name Descovy® and for PrEP in people at risk for HIV in October 2019, However, it has certain limitations, such as not being indicated for prevention in cisgender women at risk for infection through receptive vaginal intercourse [33, 34]. The CDC included TAF-FTC in its 2021 updated guidelines for PrEP and HIV treatment, highlighting its better safety profile regarding renal and bone density effects compared to TDF. However, the WHO has been more cautious in recommending TAF-FTC due to its cost and limited availability in low-resource countries, continuing to recommend TDF-FTC as the standard for PrEP in these settings [35].
The DISCOVER study, a phase III, double-blind, multicenter trial, evaluated the efficacy of TAF-FTC compared to TDF-FTC in 5387 cisgender MSM and TGW at high risk for HIV. The study found TAF-FTC to be noninferior to TDF-FTC, with an HIV incidence of 0.16 cases per 100 person-years in the TAF-FTC group compared to 0.34 in the TDF-FTC group. At 48-week follow-up, TAF-FTC showed greater safety regarding bone mineral density, with increases in BMD at the hip (0.18%) and spine (0.50%), while TDF-FTC showed decreases (-0.99% and -1.12%, respectively). Regarding renal safety, TAF-FTC had an increase in creatinine clearance (1.8%), while TDF-FTC showed a decrease (-2.3%) [36, 37].
Dapivirine Vaginal Ring
Dapivirine, a non-nucleoside reverse transcriptase inhibitor (NNRTI), was initially developed as an oral HIV treatment but was later repurposed for use as a topical microbicide [38]. The International Partnership for Microbicides (IPM) developed a vaginal ring containing dapivirine for use in HIV prevention in women in developing countries [39]. Since 2012, a ring containing dapivirine has entered phase 3 clinical trials to determine its efficacy and safety. The ASPIRE study evaluated the dapivirine vaginal ring in 2629 women from Malawi, South Africa, Uganda, and Zimbabwe, comparing it to a placebo ring [40]. The study found a 27% reduction in HIV-1 incidence in the dapivirine group (95% CI, 1 to 46; P=0.046). Excluding two low-adherence sites, efficacy increased to 37%, with the highest efficacy observed in women aged 25 years and older, reaching 61%. Similarly, the Ring Study, a phase III clinical trial with 77 women from South Africa and Uganda, found a 31% lower incidence of HIV-1 infection in the dapivirine group compared to the placebo group (HR: 0.69; 95% CI 0.49 to 0.99; p=0.04), with no significant age differences. Both studies reported no significant adverse effects related to the medication [41]. Subsequently, the open-label extension studies of the clinical trial showed that high uptake and persistence use increased the effectiveness and acceptability in the participants. The HOPE study reported an HIV-1 incidence of 2.7 per 100 person-years (95% CI 1.9–3.8, 35 infections), compared to an expected incidence of 4.4 per 100 person-years (95% CI 3.2–5.8) in the ASPIRE placebo group [42]. In addition, The DREAM study showed higher adherence compared to The Ring Study, with an HIV-1 incidence of 1.8 per 100 person-years, 62% lower than the simulated placebo rate [43]. Although dapivirine ring has received favorable opinions from the European Medicines Agency and WHO, it is not available in the USA, since IPM withdrew its FDA application due to concerns about the data not supporting approval compared to other HIV prevention options [39, 44, 45].
Cabotegravir
Cabotegravir is a long-acting integrase strand transfer inhibitor (INSTI) for HIV PrEP, which prevents HIV replication by inhibiting DNA integration. It is the first long-acting injectable for HIV PrEP and is administered every two months via intramuscular injection, offering a convenient alternative to daily oral PrEP [46, 47]. Ongoing phase 3 trials, notably HPTN 083, have evaluated Cabotegravir’s efficacy and safety. In this trial, Cabotegravir showed a 68% reduction in HIV risk compared to daily TDF/FTC in MSM and TGW. The trial also reported a favorable safety profile, with fewer participants discontinuing due to adverse events than in the TDF/FTC group [46]. The HPTN 084 trial, focused on cisgender women in sub-Saharan Africa, showed an 88% reduction in HIV risk with Cabotegravir compared to TDF/FTC [47]. To assess the long-term safety and efficacy of Cabotegravir for PrEP, the PALISADE study has started as a follow-up to HPTN 083 and 084. It monitors participants who completed these trials, continuing with long-acting Cabotegravir administration. The study focuses on evaluating new HIV infections, serious adverse events, Grade 3 and 4 injection site reactions. Results are still pending [48].
Cabotegravir was authorized by the U.S. FDA in December 2021 for use as PrEP [49]. and included in the CDC’s 2021 PrEP guidelines as a viable option for individuals at high risk of HIV who prefer less frequent dosing [18]. In 2022, the WHO included Cabotegravir in its PrEP recommendations for low-income and resource-limited settings, emphasizing its long-acting formulation that improves adherence and reduces the burden of daily medication. However, challenges such as access to testing, assay formats, and potential resistance remain for its implementation [50]. Despite its greater efficacy and acceptance in key HIV populations, Cabotegravir would require affordable generic production to ensure cost-effectiveness, considering its higher number needed to treat compared to TDF/FTC [46, 47, 51, 52]. In July 2022, ViiV Healthcare and the Medicines Patent Pool signed sublicensing agreements with Aurobindo, Cipla, and Viatris to produce generic Cabotegravir LA for HIV prevention. The agreement aims to expand access across 90 countries, particularly low-income and sub-Saharan African nations, by providing affordable long-acting PrEP options. Until generics are available, Cabotegravir LA will be offered at a non-profit price in eligible countries [53].
Real-world data confirms the effectiveness of Cabotegravir, with a 94% adherence rate in a 2023 US cohort. However, challenges persist regarding access and adherence, particularly with following CDC testing guidelines [54]. In contrast, a US community-based study found poor prescription follow-up and moderate discontinuation rates, mainly due to access issues, insurance coverage, and side effect [55].
Cabotegravir’s long-acting formulation benefits populations with challenges to daily medication adherence, such as those facing homelessness, substance use, domestic violence, or mental health issues. Its bi-monthly administration also reduces stigma and daily reminders of HIV risk, improving users’ overall quality of life [56, 57]. Studies are ongoing to evaluate the long-term safety and effectiveness of Cabotegravir for PrEP [58], as well as its potential use in combination with other long-acting agents for HIV treatment [59-61].
Lenacapavir
Lenacapavir is a long-acting capsid inhibitor developed by Gilead Sciences for treating HIV-1 infection. It is available in oral tablet and subcutaneous injection forms, with the injection administered twice yearly [62]. Lenacapavir works by targeting the HIV viral capsid, inhibiting replication at both early and late stages of the virus’s life cycle. It binds to a specific region of the capsid, preventing the virus from entering the cell’s nucleus and integrating its genetic material [63]. Approved by the FDA in 2022, Lenacapavir is used in combination with other antiretrovirals to treat adults whose HIV infections are resistant to, intolerant of, or unsafe to treat with other available therapies [64]. Additionally, Lenacapavir is currently being investigated for HIV PrEP in clinical trials known as the Purpose studies [65]. The Purpose I study, a Phase 3, multicenter, double-blind, randomized clinical trial, recently reported on the efficacy and safety of the twice-yearly Lenacapavir injection compared to the established PrEP regimens F/TAF and F/TDF, in cisgender young women from South Africa and Uganda [66]. In the Purpose I study, the primary efficacy endpoint was the incidence of HIV infection. Among the 55 total incident infections, none occurred in the Lenacapavir group (0 per 100 person-years; 95% CI, 0.00 to 0.19). In contrast, the F/TAF group had 39 infections (2.02 per 100 person-years; 95% CI, 1.44 to 2.76) and the F/TDF group had 16 infections (1.69 per 100 person-years; 95% CI, 0.96 to 2.74). In terms of safety, the most common adverse event was injection-site reactions, occurring in 68.8% of the Lenacapavir group, with subcutaneous nodules in 63.8%. All reactions were grade 1 or 2, and their occurrence decreased with subsequent doses, a typical trend in HIV treatments [67]. Other adverse events in the Lenacapavir group, such as headache, urinary tract infection, and genitourinary chlamydia, were similar to those in the other groups, with less nausea and vomiting reported in the Lenacapavir group. Regarding laboratory abnormalities, 90.5% of participants experienced abnormalities, mostly grade 1 or 2 severities. In the Lenacapavir group, 20.7% had grade 1 and 64.7% had grade 2 abnormalities, similar to the F/TAF (20.4% and 64.9%) and F/TDF (18.7% and 66.5%) groups. Recently, the preliminary results of the Purpose II study were released, showing that of 2180 participants, only two were infected with HIV, demonstrating 96% relative risk reduction. Additionally, Lenacapavir is 89% more effective than once-daily Truvada® (incidence rate ratio 0.11, p=0.00245) [68]. The WHO is awaiting the final results of the Purpose II study to with experts and stakeholders on strategies for implementing Lenacapavir as PrEP [69]. In October 2024, an agreement was made between the manufacturer and several laboratories to produce a generic version of Lenacapavir, aiming to target regions with a high incidence of HIV [70].
CURRENT STATUS OF THE HIV PrEP IMPLEMENTATION IN LATIN AMERICA
Despite its proven efficacy, the implementation and availability of PrEP vary significantly across LA, presenting a unique set of challenges and opportunities. According to recent reports, 19 out of 20 countries in the LA region have policies approving daily oral PrEP for HIV prevention, with all including at least TDF-FTC as the minimum standard for PrEP use (See Table 2). However, none of the countries in the region have approved the long-acting injectable Cabotegravir for PrEP, which the U.S. FDA approved in 2021 [49]. Brazil is currently the only LA country reviewing Cabotegravir for approval with ongoing implementation studies [71-73]. Additionally, no country in LA has approved the Dapivirine Ring or Lenacapavir for PrEP use.
Table 2 - PrEP in Latin America through 24 July 2024.
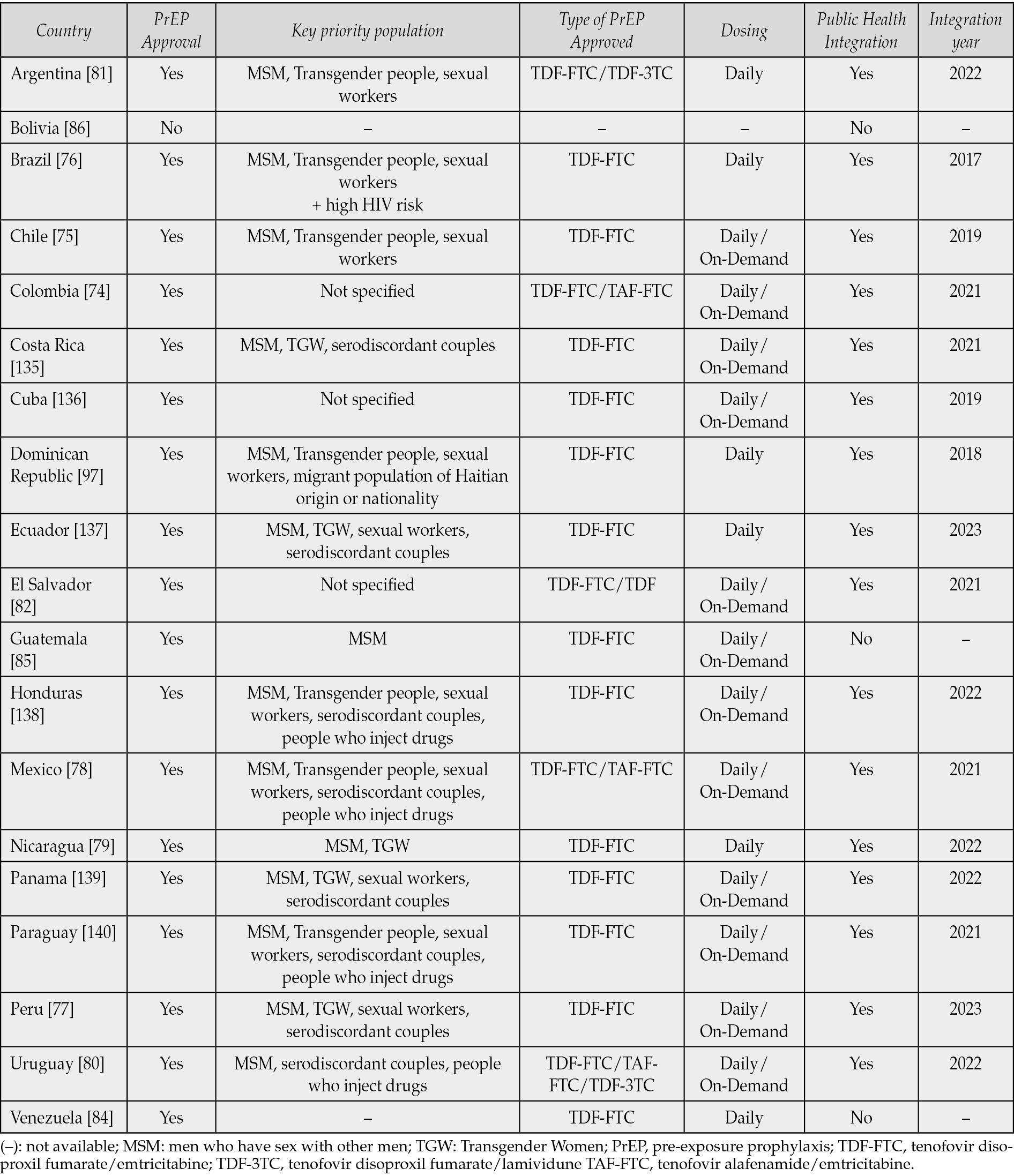
Most Latin American countries (85%) have integrated PrEP into their public health systems, making it available at no cost to key populations. This initiative involves providing medication as part of healthcare services, with monthly follow-ups to monitor adherence and assess any potential adverse effects. Brazil became the first country to provide free PrEP through its public health system to key populations in 2017. Other countries, such as Colombia, Chile, and Mexico, offer PrEP through public systems, private clinics, and non-governmental organizations (NGOs) [74-78]. PrEP was most recently approved and integrated into the public health systems of Ecuador and Peru in 2023. On the other hand, Bolivia’s status on PrEP approval and implementation remains uncertain. Nicaraguan media reports PrEP approval and integration into public health since 2022, but no official data confirms this [79].
TDF-FTC is the standard regimen for PrEP in every Latin American country. However, only 15% of these countries, including Colombia, Mexico, and Uruguay, also offer TAF-FTC as an alternative. Uruguay and Argentina have incorporated TDF-3TC into their guidelines as a PrEP option, as lamivudine (3TC) is considered an analogue of FTC. However, this regimen requires three pills (one TDF and two 3TC) raising potential concerns about adherence [80,81]. El Salvador is the only country in the region where TDF monotherapy is recommended for PrEP [82]. While all countries with PrEP approval follow a daily dosing regimen, 63% have also adopted on-demand dosing (Table 2).
Despite this public health integration, the implementation and accessibility of PrEP vary widely across the region [83]. According to the most recent data published by PAHO, there were 156,890 people on PrEP during 2023. Among the countries with the highest PrEP coverage, Brazil leads with 110,427 users, followed by Mexico with 14,108. and Colombia with 5,863 people on PrEP. Other countries such as Guatemala and Argentina have reported 4,324 and 3,927 users respectively (Figure 1) [7]. It is important to highlight the case of Brazil, Mexico, and Colombia, which have experienced a significant increase in their PrEP coverage in recent years. The number of PrEP users increased sevenfold over the last five years in Brazil; while in Mexico, a fivefold increase has been reported since PrEP was introduced to the public health system in 2021. Likewise, a ninefold increase was noted in Colombia since 2021. However, many countries still face significant barriers. For instance, Venezuela’s access to PrEP is severely restricted due to political instability, with the medication available only through a private pharmacy chain [84]. In Guatemala, access is limited to an NGO-sponsored project, while Bolivia has outdated guidelines from 2017, which do not mention PrEP [85, 86].
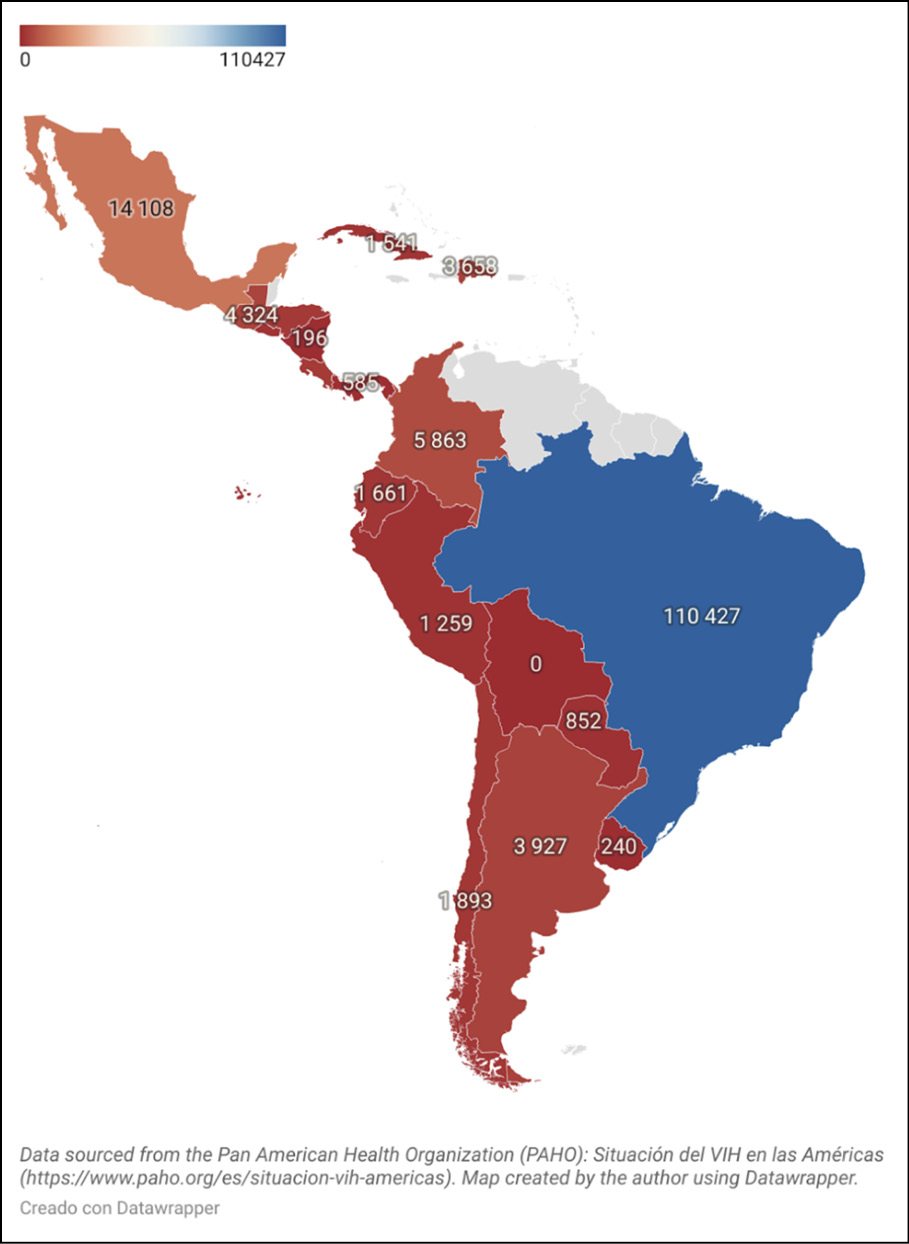
Figure 1 - Number of people on PrEP in Latin America, 2023.
Awareness plays an important role in the successful implementation of PrEP. Studies suggest that lower awareness correlates with lower rates of key populations seeking PrEP in LA countries [24]. However, other factors, such as willingness to prescribe PrEP, may also play an important role [87]. Interestingly, a higher risk of HIV infection does not seem to correlate with higher PrEP awareness. A study conducted in Brazil, Mexico, and Peru revealed inconclusive results between risk and awareness, possibly revealing a gap in public health education [88].
A recent implementation study conducted in Brazil, Mexico, and Peru revealed the feasibility of implementing same-day PrEP in LA countries, although there is limited data on long-term engagement and adherence, which may be limited by access issues, insurance coverage, or side effects. Moreover, the study observed a tendency for non-white individuals to be more prone to loss to follow-up compared to white individuals [24]. However, further research is needed to confirm this finding and to better understand the underlying factors contributing to these disparities.
While most public health systems in LA offer PrEP to high-risk individuals at no cost, the central governments obtain these medications at different prices. For example, in Chile, the cost of Truvada® has been reported to range from 200 to 450 USD per month depending on the source [89]. Ecuador is one of the few countries that has published the cost of PrEP in its national health ministry guidelines, setting the price at 4.57 USD per month for TDF-FTC [90]. In Brazil, the estimated monthly cost of PrEP with Truvada® is 22.50 USD, which equates to approximately 0.75 cents per pill [91,92]. The PAHO Strategic Fund seeks to reduce the cost of PrEP by providing a one-month supply of TDF-FTC for 4.50 USD to various LA countries to date. However, evaluating its impact is challenging due to the limited public information available on the specific medications procured through this initiative [93].
Many LA countries do not provide sufficient specifics on PrEP availability, usage, and policy plans. For instance, Bolivia initially approved Truvada® but later withdrew the approval [94]. During the period when Truvada® was approved, there was no accessible data on its availability. Similarly, while Colombia has included Cabotegravir in its 2023 PrEP guidelines, it has not yet received official approval from the National Food and Drug Administration and has no ongoing implementation studies [74].
Although UNAIDS and PAHO provide the most current PrEP data for LA [3,7], centralized sources such as PrEP Watch and AIDS Vaccine Advocacy Coalition often lack detailed information on PrEP policies in LA compared to regions like the Global North and Sub-Saharan Africa [95, 96]. This information gap is crucial as it impedes a thorough understanding of PrEP implementation and access in the region.
It is worth mentioning that 76% of Latin American countries with approved PrEP have published clinical guidelines that identify key populations who may benefit from its use, such as MSM, TGW, sex workers, serodiscordant couples, and people who inject drugs. These guidelines, published between 2018 and 2023, vary in terms of indication and eligibility. For instance, some countries like Ecuador define ‘substantial risk’ with clear criteria, while the Dominican Republic broadens its scope to include migrants from Haiti, reflecting the high HIV burden in this population [97]. In contrast, some countries adopt more flexible language, allowing for individualized assessments for those at risk of HIV exposure who may not belong to a key population. Notably, all guidelines emphasize that PrEP is intended for individuals at high risk of HIV, even when specific populations are not explicitly defined.
Despite significant progress in approving and implementing PrEP across LA, several challenges persist in ensuring widespread and equitable access. These include inconsistent availability, inadequate education for both patients and healthcare providers, and the need for more detailed and comprehensive national guidelines. Barriers to PrEP implementation in Latin America include inconsistent clinical guidelines with varying criteria for key populations and risk behaviors, creating confusion for both healthcare providers and patients. While some countries have integrated PrEP into their public health systems, others have not, limiting access, especially for low-income populations in places like Guatemala, Venezuela, and Bolivia. Although Brazil has included PrEP in its public health system, access remains limited in rural areas, and Colombia’s guidelines do not specify priority populations for PrEP [14]. In Mexico, a qualitative study found that barriers to PrEP access include limited awareness, stigma, economic challenges, geographic distance from healthcare centers, long wait times, and misinformation, all of which hinder access [98]. Unlike Global North and Sub-Saharan Africa, Latin America lacks centralized PrEP information, such as the data provided by platforms like PrEPWatch. This underscores the need for similar resources in Latin America to improve awareness and ensure equitable access to PrEP for both healthcare providers and the public [99]. Additionally, institutional barriers, such as fragmented healthcare systems, long distances to PrEP clinics, logistical issues related to pill distribution, and resistance from healthcare professionals to prescribe PrEP, exacerbate the situation [100]. While national policies exist, they often lack the specificity and clarity needed for effective implementation, especially regarding transparent data on how many people are receiving PrEP, including key populations. Strengthening these guidelines and improving access to information is crucial to fully leverage PrEP as a vital tool in HIV prevention across the region.
While Latin America has made progress in implementing PrEP, its effectiveness in reducing HIV transmission has been demonstrated globally, with significant impacts in countries like the U.S., the UK, South Africa, and Australia, among others. In the United States, the number of PrEP users has steadily increased, with a notable 17% rise from 2022 to 2023, reflecting growing awareness and acceptance since its introduction in 2012 [101]. The UK’s PrEP Impact Trial (2017-2020), involving over 24,000 participants, found that PrEP reduced HIV risk by 86%, leading to its routine use in England starting in October 2020 [102]. Similarly, South Africa, which has the largest population of people living with HIV, introduced PrEP in 2016. By 2020, access to PrEP was expanded as part of the ‘combination prevention’ approach, a comprehensive strategy that includes behavioral, biomedical, and structural HIV prevention programs. This approach has significantly reduced HIV incidence, especially among high-risk groups such as young women [103]. In Australia, where PrEP has been subsidized since April 2018, a recent cohort study found that individuals with at least 60% adherence to PrEP experienced a 78.5% reduction in HIV incidence [104]. Like Latin America, these countries also encounter challenges in PrEP implementation and success, such as stigma, logistical obstacles, and adherence difficulties. Unfortunately, these barriers are pervasive and affect PrEP adoption and effectiveness globally.
Regarding stigma associated with adherence to PrEP, studies on this topic are scarce in Latin America. There is limited research in the region specifically focusing on PrEP and stigma. Outside the region, in one qualitative study, the majority of participants reported that PrEP users continue to be associated with derogatory terms. These terms contribute to the stigma surrounding PrEP use and may discourage individuals from seeking or continuing PrEP [105]. In another study surveying the MSM population, stigma related to PrEP was strongly associated with a lack of interest in using it. Specifically, participants who believed that PrEP is only for people who engage in high-risk sexual behavior (e.g., “promiscuous” individuals) were less likely to express interest in using it [106]. PrEP-related stigma has become a significant social issue, reported by research participants from various trial sites, different populations, and across multiple continents. Social marketing efforts need to address PrEP-related stigma through health promotion campaigns for HIV-affected communities and individuals at high risk of HIV who could benefit from access to PrEP [107].
COST-EFFECTIVENESS STUDIES REGARDING PrEP IN LATIN AMERICA
Cost-effectiveness analyses have become crucial in determining the feasibility and potential impact of PrEP across different subpopulations across LA. The uptake of PrEP is steadily increasing in the region, with implementation projects in countries such as Brazil, Mexico, and Peru [108]. One analysis suggested that PrEP implementation could significantly reduce both associated healthcare costs over time. This study emphasized the importance of targeting high-risk groups to maximize the economic benefits of PrEP programs [109]. In this regard, having detailed and up-to-date cost data is crucial for accurately assessing the cost-effectiveness of large-scale delivery models, as well as for developing effective strategies that ensure effective service integration [110].
PrEP cost-effectiveness programs rely on the ability to integrate into the existing healthcare system and the ability to generate appropriate demand among key populations [14]. Brazil has made notable progress in this regard, with fifty-six cities now offering PrEP through the public health system [108]. A study conducted in Brazil [91] analyzed the cost-effectiveness of PrEP for HIV prevention among MSM and TGW and found that daily PrEP with tenofovir/emtricitabine would be highly cost-effective and increase life expectancy. The study also showed a significant impact of PrEP, reducing the lifetime risk of HIV infection from 50.5% to 40.1% while promoting early detection of HIV cases. Additionally, the incremental cost-effectiveness ratio (ICER) was calculated at 2530 dollars per year of life saved, reinforcing PrEP as a highly cost-effective intervention [91].
A study conducted in Peru focused on TGW found that a combination of interventions, including increased condom use, expanded antiretroviral treatment coverage, and PrEP, could prevent 47% of new HIV infections over ten years with a cost-effectiveness ratio of 509 dollars per disability-adjusted life year (DALY) averted [111]. Similarly, a systematic review demonstrated that these combined interventions significantly reduced HIV transmission rates, with effectiveness estimates ranging from 30% to 90%, depending on adherence and coverage [112]. Lastly, a study in MSM and TGW in Lima-Peru, showed that with a relatively low PrEP coverage (5%), over 8% of new infections could be prevented if higher-risk individuals were prioritized (TGW and sex workers), and their adherence levels were ensured [113]. Therefore, it is critical to target specific at-risk populations to achieve high coverage and adherence to these interventions.
While these studies highlight the potential of PrEP in reducing HIV transmission, there is a need for comprehensive cost-benefit analyses that also account for indirect benefits, such as improved quality of life and reduced healthcare costs. The purchasing and negotiation for antiretrovirals could reduce costs and significantly improve the cost-effectiveness of PrEP [113]. Additionally, future research should explore the influence of social and behavioral factors on PrEP adherence and effectiveness, as well as the long-term sustainability of these interventions.
PrEP and sexually transmitted diseases
Sexually transmitted diseases (STDs) represent a critical concern in the context of HIV pre-exposure prophylaxis (PrEP) use due to their potential to significantly increase the risk of HIV transmission, thereby underscoring the importance of systematic monitoring and control among PrEP users [114]. The integration of STD services into PrEP programs is essential, particularly for high-risk populations, where the prevalence of STDs can be significantly higher and may compromise the overall effectiveness of HIV prevention strategies [115].
Systematic reviews have shown that among MSM using PrEP, STD prevalence before starting PrEP was alarmingly high, with rates of 12% for gonorrhea, 11% for chlamydia, and 5% for syphilis, far exceeding global prevalence rates of 0.7%, 2.5%, and 0.6%, respectively [116]. These figures highlight the imperative of embedding STD monitoring into PrEP programs to mitigate transmission risks, improve treatment outcomes, and strengthen the overall impact of HIV prevention initiatives.
Countries like Perú and Brazil have adopted comprehensive strategies to integrate STD monitoring into their PrEP programs. In Brazil, the guidelines for PrEP emphasize routine STD screening as part of the initial and ongoing clinical and laboratory evaluations [76]. Similarly, Peru’s PrEP implementation integrates STD screening and treatment into its prevention efforts. The Peruvian Ministry of Health (MINSA) mandates regular testing for HIV and other STDs, complemented by counseling and the provision of condoms and lubricants to minimize risks. Both countries emphasize periodic follow-ups and education on adherence to PrEP to address infections that PrEP does not prevent [77].
While integrating STD screening into PrEP programs has undeniable health benefits, it presents significant cost-effectiveness challenges in both organizational and financial terms. Comprehensive testing, including rapid diagnostic tests (RDTs) and molecular testing, requires substantial investment in laboratory infrastructure, staff training, and operational costs [112, 117]. For example, integrating STD testing into Brazil’s program was estimated to add an average cost of $25 per test per individual over two years, increasing overall program expenses by approximately 50-60% and highlighting the significant financial burden such services can impose [118].
Even though costs are particularly prohibitive in resource-limited settings like some countries in Latin America, targeted strategies can enhance efficiency and affordability. The use of dual HIV/syphilis RDTs, for instance, is both cost-effective and impactful, expanding testing coverage while minimizing budgetary constraints [116]. By addressing these cost limitations and leveraging available resources effectively, PrEP programs can integrate STD services to maximize health outcomes while advancing global public health goals.
INTERVENTIONS TO IMPROVE PrEP COVERAGE IN LATIN AMERICA
Several studies have identified strategies to improve adherence and coverage, addressing barriers such as stigma and misinformation, while expanding access in at-risk populations to reduce HIV incidence [119].
A scoping review conducted in 2021 highlights different strategies for PrEP implementation that have helped improve coverage across different countries [14]. Among the various interventions analyzed, the ImPrEP demonstration project, conducted in Brazil, Mexico, and Peru, showed that same-day PrEP initiation is an effective strategy to enhance treatment adherence and early continuation, with a positive impact on increasing coverage [87]. However, the adherence to PrEP in Latin America varies significantly among different population groups. The ImPrEP study [24], which included 9,509 participants from Brazil, Peru and Mexico, found that 68.1% of participants adhered to PrEP, and of these, 70.3% maintained long-term adherence. However, the odds of adherence were lower among transgender women compared to cisgender men [24]. In addition, a study in Peru identified specific barriers to PrEP adherence among men who have sex with men (MSM) and transgender women, including structural and social challenges that affect continued PrEP use [120]. These findings underscore the need to implement comprehensive and culturally tailored strategies that address gender disparities and other factors influencing PrEP adherence in the region.
Innovative PrEP strategies, such as long-acting injectables, vaginal rings, and monthly pills, show promise in overcoming adherence barriers [14]. While traditional oral PrEP is effective, it faces challenges like daily pill burden, stigma, and limited healthcare access. Studies on long-acting options demonstrate improved adherence and HIV prevention efficacy [121]. However, no Latin American country has widely approved these methods, largely due to infrastructure, training, and cost barriers. Ongoing trials in Peru, Brazil, and Argentina will provide valuable insights for future implementation [14].
Event-driven PrEP (ED-PrEP) is an alternative strategy aimed at improving adherence, particularly among MSM, as recommended by the WHO in 2019 [14, 29]. The AMPrEP study demonstrated high adherence to ED-PrEP, with most participants achieving good or excellent protection against HIV during condomless anal sex (CAS) [122]. Notably, 90.3% of CAS acts with unknown casual partners were protected. Furthermore, over 1,046 three-month periods of ED-PrEP use, 81.4% of users demonstrated consistently correct PrEP use, indicating strong long-term adherence. Although several studies have shown that ED-PrEP is highly effective in preventing HIV, research on this strategy remains limited and is predominantly conducted among individuals with higher education levels [122].
Biobehavioral interventions have proven essential in improving PrEP coverage and adherence among key populations in LA. The integration of mobile health applications and digital tools has significantly enhanced adherence by offering continuous counseling, risk assessments, and social support. In Brazil, the PrEP Brasil Study demonstrated a high adherence rate, with 74% of participants consistently taking daily oral FTC/TDF. The study also reported an overall reduction in HIV risk, as evidenced by a decrease in the mean number of sexual partners from 11.4 at the beginning of the study to 8.3 by its conclusion. Similarly, the PrEP Brasil Demonstration Project found that 78.5% of participants maintained protective drug levels, reflecting robust adherence to PrEP. In Peru, the TransPrEP Study, which focused on promoting PrEP uptake through individual counseling and group workshops for TGW, revealed that 80% of participants in the intervention group had detectable tenofovir levels in their hair samples, compared to only 51.1% in the control group. Across both Brazil and Peru, adherence to PrEP and retention within intervention programs were notably high, with approximately 74% to 80% of participants taking PrEP at least four times per week, alongside a reduction in risky sexual behaviors [123].
Results indicate that these interventions are effective in increasing awareness and adherence to PrEP, with reported adherence rates as high as 78.5% [124]. However, a lack of implementation is observed in several countries in the region where HIV rates are high, underscoring the need for a more comprehensive and structural approach to address challenges in HIV prevention, including stigma and discrimination towards key populations [108, 113].
While there is a high demand for PrEP, guaranteeing access to PrEP for everyone remains challenging globally. Especially in low-middle income countries, such as those in Latin America, resource constraints and structural obstacles generate greater delays in optimal healthcare delivery than those seen in high-income countries [125]. A notable innovation in the region is Brazil, where telehealth for PrEP was tested between 2019 and 2020, with 52% of users opting for remote consultations, reducing PrEP discontinuation by 34% and maintaining similar adherence to in-person care [126]. Other innovative approaches to HIV PrEP delivery are emerging in Latin America, though many challenges remain and have yet to be fully documented in the literature.
PrEP is often delivered in a centralized way in a clinical or hospital setting that include specialized clinics, reproductive health services and study centers. Meanwhile there are also community-based models that include community health centers, pharmacies and primary care practitioner practice and home-based models [127]. A study in South Africa evidenced that PrEP initiation at a pharmacy or mobile clinic was valued equally to initiating PrEP at a clinic, but a community pop-up site was less preferred [128]. This same study showed that participants preferred collecting PrEP from a pharmacy (aOR = 5.02, 95% CI = 3.45–7.31), through home delivery (aOR = 2.18, 95% CI = 1.26–3.78) and from a vending machine (aOR = 1.43, 95% CI = 1.02–1.99) relative to where they initiated PrEP. Therefore, there is a need to expand decentralized and self-led HIV prevention services to meet users’ preferences.
Novel pick-up points such as the use of community-based PrEP delivery methods, mobile clinics, tents and home delivery may be key to reach vulnerable populations who are often marginalized and at high risk of contracting HIV [125]. Nonetheless, the innovative digital options of delivering PrEP such as mHealth and telemedicine approaches are only being applied in a minority of patients and mainly high-resource settings [127].
CONCLUSIONS
In conclusion, PrEP has emerged as a vital strategy for HIV prevention in Latin America, yet its implementation varies significantly across the region. While countries like Brazil and Argentina have made notable advancements in providing access to PrEP through public healthcare systems, challenges remain, particularly regarding integration and coverage. Despite a high willingness to use PrEP among potential users, barriers such as a lack of national treatment guidelines, stigma, and misinformation hinder broader uptake.
Innovative strategies are needed to expand PrEP options for high-risk populations, including long-acting formulations like injectable antiretrovirals and vaginal rings, which have shown promise in enhancing adherence and increasing coverage. To maximize the impact of this crucial preventive tool, continued research, and targeted interventions are essential to address existing barriers and ensure that all at-risk populations can benefit from PrEP. The future of HIV prevention in the region relies on sustained efforts to enhance awareness, accessibility, and adherence to PrEP among vulnerable communities.
Conflict of interest
None.
Funding
This research received no external funding.
REFERENCES
[1] WHO. The Global Health Observatory. HIV. World Health Organization 2024. https://www.who.int/data/gho/data/themes/hiv-aids/hiv-aids (accessed October 21, 2024).
[2] WHO. Global health sector strategies on, respectively, HIV, viral hepatitis and sexually transmitted infections for the period 2022-2030. World Health Organization 2022. https://www.who.int/publications/i/item/9789240053779 (accessed October 21, 2024).
[3] UNAIDS. Global AIDS Strategy 2021-2026 - End Inequalities. End AIDS. The Joint United Nations Programme on HIV/AIDS 2021. https://www.unaids.org/es/Global-AIDS-Strategy-2021-2026 (accessed October 21, 2024).
[4] Fondo Mundial. Luchar contra las pandemias y construir un mundo más saludable y equitativo. Estrategia del Fondo Mundial (2023-2028). The Global Fund 2021. https://www.theglobalfund.org/media/11692/strategy_globalfund2023-2028_narrative_es.pdf (accessed October 21, 2024).
[5] Castor D, Heck CJ, Quigee D, Telrandhe NV, Kui K, Wu J, et al. Implementation and resource needs for long-acting PrEP in low- and middle-income countries: a scoping review. J Int AIDS Soc. 2023; 26: e26110. https://doi.org/10.1002/jia2.26110.
[6] UNAIDS. The Urgency of Now. Aids at a Crossroads. Joint United Nations Programme on HIV/AIDS 2024. https://www.unaids.org/sites/default/files/media_asset/2024-unaids-global-aids-update_en.pdf (accessed October 21, 2024).
[7] PAHO. HIV Situation in the Americas. Data on PrEP, HIV diagnosis and treatment in Latin America and the Caribbean. Pan American Health Organization 2023. https://www.paho.org/en/hiv-situation-americas (accessed October 21, 2024).
[8] WHO. HIV and AIDS. World Health Organization 2024. https://www.who.int/news-room/fact-sheets/detail/hiv-aids?gad_source=1&gclid=CjwKCAjw8rW2BhAgEiwAoRO5rMPiewlo4_6LlccY66EeK_L6oeigWEJXXSshKpVQgvSNcNeT9KqI2xoC6IMQAvD_BwE (accessed October 21, 2024).
[9] Huynh K, Vaqar S, Gulick. PG. HIV Prevention. StatPearls [Internet] 2024. https://www.ncbi.nlm.nih.gov/books/NBK470281/ (accessed October 21, 2024).
[10] Sullivan PS, Mena L, Elopre L, Siegler AJ. Implementation Strategies to Increase PrEP Uptake in the South. Curr HIV/AIDS Rep. 2019; 16: 259-269. https://doi.org/10.1007/s11904-019-00447-4.
[11] Bavinton BR, Grulich AE. HIV pre-exposure prophylaxis: scaling up for impact now and in the future. The Lancet Public Health. 2021; 6: e528-e533. https://doi.org/10.1016/S2468-2667(21)00112-2.
[12] Kuman Tunçel Ö, Taşbakan SE, Gökengin D, et al. The deep impact of the COVID-19 pandemic on medical students: An online cross-sectional study evaluating Turkish students’ anxiety. Int J Clinical Practice. 2021; 75: e14139. https://doi.org/10.1111/ijcp.14139.
[13] Zhao R, Fairley CK, Cook AR, Phanuphak N, He S, Tieosapjaroen W, et al. Optimising HIV pre-exposure prophylaxis and testing strategies in men who have sex with men in Australia, Thailand, and China: a modelling study and cost-effectiveness analysis. The Lancet Global Health. 2024; 12: e243-e256. https://doi.org/10.1016/S2214-109X(23)00536-3.
[14] Murphy L, Bowra A, Adams E, Cabello R, Clark JL, Konda K, et al. PrEP policy implementation gaps and opportunities in Latin America and the Caribbean: a scoping review. Ther Adv Infect Dis. 2023; 10.20499361231164030. https://doi.org/10.1177/20499361231164030.
[15] Plosker GL. Emtricitabine/Tenofovir Disoproxil Fumarate: A Review of its Use in HIV-1 Pre-Exposure Prophylaxis. Drugs. 2013; 73: 279-291. https://doi.org/10.1007/s40265-013-0024-4.
[16] U.S. Food & Drug Administration. Drug Approval Package. Truvada (Emtricitabine and Tenofovir Disoproxil Fumarate) Tablets. Access Data FDA 2006. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2004/021752s000_TruvadaTOC.cfm (accessed October 21, 2024).
[17] HIV.gov. FDA approves first drug for reducing the risk of sexually acquired HIV infection. HIVGov 2012. https://www.hiv.gov/blog/fda-approves-first-drug-for-reducing-the-risk-of-sexually-acquired-hiv-infection (accessed October 21, 2024).
[18] US Public Health Service. Preexposure Prophylaxis for the Prevention of HIV Infection in the United States – 2021 Update Clinical Practice Guideline. Centers for Disease Control and Prevention 2021. https://www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf (accessed October 21, 2024).
[19] WHO. Access to antiretroviral drugs in low- and middleincome countries. World Health Organization 2014. https://new.aidsdatahub.org/sites/default/files/resource/who-technical-report-access-antiretroviral-drugs-low-middle-income-countries.pdf (accessed October 21, 2024).
[20] Baeten JM, Donnell D, Ndase P, Mugo NR, Campbell JD, Wangisi J, et al. Antiretroviral prophylaxis for HIV prevention in heterosexual men and women. N Engl J Med. 2012; 367: 399-410. https://doi.org/10.1056/NEJMoa1108524.
[21] Grant RM, Lama JR, Anderson PL, McMahan V, Liu AY, Vargas L, et al. Preexposure Chemoprophylaxis for HIV Prevention in Men Who Have Sex with Men. N Engl J Med. 2010; 363: 2587-2599. https://doi.org/10.1056/NEJMoa1011205.
[22] Choopanya K, Martin M, Suntharasamai P, et al. Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomised, double-blind, placebo-controlled phase 3 trial. The Lancet. 2013; 381: 2083-2090. https://doi.org/10.1016/S0140-6736(13)61127-7.
[23] Liu AY, Cohen SE, Vittinghoff E, et al. Preexposure Prophylaxis for HIV Infection Integrated With Municipal- and Community-Based Sexual Health Services. JAMA Intern Med. 2016; 176: 75. https://doi.org/10.1001/jamainternmed.2015.4683.
[24] Veloso VG, Cáceres CF, Hoagland B, et al. Same-day initiation of oral pre-exposure prophylaxis among gay, bisexual, and other cisgender men who have sex with men and transgender women in Brazil, Mexico, and Peru (ImPrEP): a prospective, single-arm, open-label, multicentre implementation study. The Lancet HIV. 2023; 10: e84-e96. https://doi.org/10.1016/S2352-3018(22)00331-9.
[25] Mulligan K, Glidden DV, Anderson PL, et al. Effects of Emtricitabine/Tenofovir on Bone Mineral Density in HIV-Negative Persons in a Randomized, Double-Blind, Placebo-Controlled Trial. Clin Infect Dis. 2015; 61: 572-580. https://doi.org/10.1093/cid/civ324.
[26] Tang EC, Vittinghoff E, Anderson PL, Cohen SE, Doblecki-Lewis S, Bacon O, et al. Changes in Kidney Function Associated With Daily Tenofovir Disoproxil Fumarate/Emtricitabine for HIV Preexposure Prophylaxis Use in the United States Demonstration Project. J Acquir Immune Defic Syndr. 2018; 77: 193-198. https://doi.org/10.1097/QAI.0000000000001566.
[27] Gandhi RT, Bedimo R, Hoy JF, Landovitz RJ, Smith DM, Eaton EF, et al. Antiretroviral Drugs for Treatment and Prevention of HIV Infection in Adults: 2022 Recommendations of the International Antiviral Society-USA Panel. JAMA. 2023; 329: 63-84. https://doi.org/10.1001/jama.2022.22246.
[28] WHO. Differentiated and simplified pre-exposure prophylaxis for HIV prevention: update to WHO implementation guidance. World Health Organization 2022. https://www.who.int/publications/i/item/9789240053694 (accessed October 21, 2024).
[29] WHO. What’s the 2+1+1? Event-driven oral pre-exposure prophylaxis to prevent hiv for men who have sex with men: update to who’s recommendation on oral prep. World Health Organization 2019. https://iris.who.int/bitstream/handle/10665/325955/WHO-CDS-HIV-19.8-eng.pdf (accessed October 21, 2024).
[30] Molina J-M, Capitant C, Spire B, et al. On-Demand Preexposure Prophylaxis in Men at High Risk for HIV-1 Infection. N Engl J Med. 2015; 373: 2237-2246. https://doi.org/10.1056/NEJMoa1506273.
[31] Molina J-M, Ghosn J, Assoumou L, et al. Daily and on-demand HIV pre-exposure prophylaxis with emtricitabine and tenofovir disoproxil (ANRS PREVENIR): a prospective observational cohort study. Lancet HIV. 2022; 9: e554-e562. https://doi.org/10.1016/S2352-3018(22)00133-3.
[32] Ruane PJ, DeJesus E, Berger D, et al. Antiviral activity, safety, and pharmacokinetics/pharmacodynamics of tenofovir alafenamide as 10-day monotherapy in HIV-1-positive adults. J Acquir Immune Defic Syndr. 2013; 63: 449-455. https://doi.org/10.1097/QAI.0b013e3182965d45.
[33] Stewart J. Descovy FDA Approval History. DrugsCom 2019. https://www.drugs.com/history/descovy.html (accessed October 21, 2024).
[34] Drugs.com. FDA Approves Descovy (emtricitabine and tenofovir alafenamide) for HIV Pre-Exposure Prophylaxis (PrEP). DrugsCom 2019. https://www.drugs.com/newdrugs/fda-approves-descovy-emtricitabine-tenofovir-alafenamide-hiv-pre-exposure-prophylaxis-prep-5068.html (accessed October 21, 2024).
[35] WHO. Pre-exposure prophylaxis (PrEP). Global HIV Programme. World Health Organization n.d. https://www.who.int/teams/global-hiv-hepatitis-and-stis-programmes/hiv/prevention/pre-exposure-prophylaxis (accessed October 21, 2024).
[36] Mayer KH, Molina J-M, Thompson MA, et al. Emtricitabine and tenofovir alafenamide vs emtricitabine and tenofovir disoproxil fumarate for HIV pre-exposure prophylaxis (DISCOVER): primary results from a randomised, double-blind, multicentre, active-controlled, phase 3, non-inferiority trial. Lancet. 2020; 396: 239-254. https://doi.org/10.1016/S0140-6736(20)31065-5.
[37] Pilkington V, Hughes SL, Pepperrell T, et al. Tenofovir alafenamide vs. tenofovir disoproxil fumarate: an updated meta-analysis of 14 894 patients across 14 trials. AIDS. 2020; 34: 2259-2268. https://doi.org/10.1097/QAD.0000000000002699.
[38] Baeten JM, Hendrix CW, Hillier SL. Topical Microbicides in HIV Prevention: State of the Promise. Annu Rev Med. 2020; 71: 361-377. https://doi.org/10.1146/annurev-med-090518-093731.
[39] International Partnership for Microbicides. IPM Statement on US Food and Drug Administration Review of Dapivirine Vaginal Ring. International Partnership for Microbicides 2021. https://web.archive.org/web/20240321005041/https:/www.ipmglobal.org/content/ipm-statement-us-food-and-drug-administration-review-dapivirine-vaginal-ring (accessed October 21, 2024).
[40] Baeten JM, Palanee-Phillips T, Brown ER, et al. Use of a Vaginal Ring Containing Dapivirine for HIV-1 Prevention in Women. N Engl J Med. 2016; 375: 2121-2132. https://doi.org/10.1056/NEJMoa1506110.
[41] Nel A, Van Niekerk N, Kapiga S, et al. Safety and Efficacy of a Dapivirine Vaginal Ring for HIV Prevention in Women. N Engl J Med. 2016; 375: 2133-2143. https://doi.org/10.1056/NEJMoa1602046.
[42] Baeten JM, Palanee-Phillips T, Mgodi NM, et al. Safety, uptake, and use of a dapivirine vaginal ring for HIV-1 prevention in African women (HOPE): an open-label, extension study. The Lancet HIV. 2021; 8: e87-e95. https://doi.org/10.1016/S2352-3018(20)30304-0.
[43] Nel A, Van Niekerk N, Van Baelen B, et al. Safety, adherence, and HIV-1 seroconversion among women using the dapivirine vaginal ring (DREAM): an open-label, extension study. The Lancet HIV. 2021; 8: e77-e86. https://doi.org/10.1016/S2352-3018(20)30300-3.
[44] European Medicines Agency. Dapivirine Vaginal Ring 25 mg - opinion on medicine for use outside EU. European Medicines Agency 2021. https://www.ema.europa.eu/en/opinion-medicine-use-outside-EU/human/dapivirine-vaginal-ring-25-mg (accessed October 21, 2024).
[45] WHO. WHO recommends the dapivirine vaginal ring as a new choice for HIV prevention for women at substantial risk of HIV infection. World Health Organization 2021. https://www.who.int/news/item/26-01-2021-who-recommends-the-dapivirine-vaginal-ring-as-a-new-choice-for-hiv-prevention-for-women-at-substantial-risk-of-hiv-infection (accessed October 21, 2024).
[46] Landovitz RJ, Donnell D, Clement ME, et al. Cabotegravir for HIV Prevention in Cisgender Men and Transgender Women. N Engl J Med. 2021; 385: 595-608. https://doi.org/10.1056/NEJMoa2101016.
[47] Delany-Moretlwe S, Hughes JP, Bock P, et al. Cabotegravir for the prevention of HIV-1 in women: results from HPTN 084, a phase 3, randomised clinical trial. The Lancet. 2022; 399: 1779-1789. https://doi.org/10.1016/S0140-6736(22)00538-4.
[48] Veeva Systems. Long-term Follow-up of Long-acting Cabotegravir (CAB LA) for PrEP (Pre-exposure Prophylaxis) in Participants at Risk of Acquiring HIV (Human Immunodeficiency Virus) (PALISADE). CTV 2024. https://ctv.veeva.com/study/long-term-follow-up-of-long-acting-cabotegravir-cab-la-for-prep-pre-exposure-prophylaxis-in-part (accessed October 21, 2024).
[49] U.S. Food & Drug Administration. FDA Approves First Injectable Treatment for HIV Pre-Exposure Prevention. FDA 2021. https://www.fda.gov/news-events/press-announcements/fda-approves-first-injectable-treatment-hiv-pre-exposure-prevention (accessed October 21, 2024).
[50] WHO. Guidelines on long-acting injectable cabotegravir for HIV prevention. World Health Organization 2022. https://www.who.int/publications/i/item/9789240054097 (accessed October 21, 2024).
[51] Pepperrell T, Cross S, Hill A. Cabotegravir—Global Access to Long-Acting Pre-exposure Prophylaxis for HIV. Open Forum Infectious Diseases. 2023; 10: ofac673. https://doi.org/10.1093/ofid/ofac673.
[52] Jamieson L, Johnson LF, Nichols BE, et al. Relative cost-effectiveness of long-acting injectable cabotegravir versus oral pre-exposure prophylaxis in South Africa based on the HPTN 083 and HPTN 084 trials: a modelled economic evaluation and threshold analysis. The Lancet HIV. 2022; 9: e857-e867. https://doi.org/10.1016/S2352-3018(22)00251-X.
[53] GSK. Medicines Patent Pool signs sublicences with Aurobindo, Cipla and Viatris to produce generic versions of ViiV Healthcare’s innovative long-acting HIV prevention medicine. GSK 2023. https://www.gsk.com/en-gb/media/press-releases/medicines-patent-pool-signs-sublicences-with-aurobindo-cipla-and-viatris-to-produce-generic-versions-of-viiv-healthcare-s-innovative-long-acting-hiv-prevention-medicine/ (accessed October 21, 2024).
[54] Mayer KH, Frick AJ, Brown C, et al. Real-World Use of Cabotegravir Long-Acting for Preexposure Prophylaxis: Trio Health Cohort. Conference on Retroviruses and Opportunistic Infections 2024. https://www.croiconference.org/abstract/real-world-use-of-cabotegravir-long-acting-for-preexposure-prophylaxis-trio-health-cohort/ (accessed October 21, 2024).
[55] Altamirano JA, Shukla P, Barnett SK. 1531. Early Real-World Experience of Long-Acting Cabotegravir (CAB) for HIV Pre-Exposure Prophylaxis (PrEP) in a Large Community-Based Clinic Network (CAN Community Health): Utilization and PrEP Persistence. Open Forum Infectious Diseases 2023; 10: ofad500.1366. https://doi.org/10.1093/ofid/ofad500.1366.
[56] Velloza J, Khoza N, Scorgie F, et al. The influence of HIV-related stigma on PrEP disclosure and adherence among adolescent girls and young women in HPTN 082: a qualitative study. J Intern AIDS Soc. 2020; 23: e25463. https://doi.org/10.1002/jia2.25463.
[57] Pillay D, Stankevitz K, Lanham M, et al. Factors influencing uptake, continuation, and discontinuation of oral PrEP among clients at sex worker and MSM facilities in South Africa. PLoS ONE. 2020; 15: e0228620. https://doi.org/10.1371/journal.pone.0228620.
[58] Landovitz RJ, Hanscom BS, Clement ME, et al. Efficacy and safety of long-acting cabotegravir compared with daily oral tenofovir disoproxil fumarate plus emtricitabine to prevent HIV infection in cisgender men and transgender women who have sex with men 1 year after study unblinding: a secondary analysis of the phase 2b and 3 HPTN 083 randomised controlled trial. The Lancet HIV. 2023; 10: e767-e778. https://doi.org/10.1016/S2352-3018(23)00261-8.
[59] John M, Williams L, Nolan G, Bonnett M, Castley A, Nolan D. Real-world use of long-acting cabotegravir and rilpivirine: 12-month results of the inJectable Antiretroviral therapy feasiBility Study ( JABS ). HIV Medicine. 2024; 25: 935-945. https://doi.org/10.1111/hiv.13647.
[60] Hsu RK, Sension M, Fusco JS, et al. 1028. Real-World Use of Long-Acting Cabotegravir + Rilpivirine in People with HIV with Detectable Viral Loads at Initiation: Findings from the OPERA® Cohort. Open Forum Infec Dis. 2023; 10: ofad500.059. https://doi.org/10.1093/ofid/ofad500.059.
[61] Thoueille P, Saldanha SA, Schaller F, et al. Real-world trough concentrations and effectiveness of long-acting cabotegravir and rilpivirine: a multicenter prospective observational study in Switzerland. The Lancet Regional Health - Europe. 2024; 36: 100793. https://doi.org/10.1016/j.lanepe.2023.100793.
[62] Paik J. Lenacapavir: First Approval. Drugs 2022; 82: 1499-1504. https://doi.org/10.1007/s40265-022-01786-0.
[63] Hitchcock AM, Kufel WD, Dwyer KAM, Sidman EF. Lenacapavir: A novel injectable HIV-1 capsid inhibitor. International Journal of Antimicrobial Agents 2024; 63: 107009. https://doi.org/10.1016/j.ijantimicag.2023.107009.
[64] U.S. Food & Drug Administration. Novel Drug Approvals for 2022. FDA 2022. https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2022 (accessed October 21, 2024).
[65] PURPOSE. PrEP Studies Overview. PURPOSE n.d. https://www.purposestudies.com/ (accessed October 21, 2024).
[66] Bekker L-G, Das M, Abdool Karim Q, Ahmed K, Batting J, Brumskine W, et al. Twice-Yearly Lenacapavir or Daily F/TAF for HIV Prevention in Cisgender Women. N Engl J Med. 2024; 391: 1179-1192. https://doi.org/10.1056/NEJMoa2407001.
[67] Kumar P, Gupta S, Segal-Maurer S, Ogbuagu O, McDonald C, Brinson C, et al. Injection site reaction experience in clinical studies of people using lenacapavir for HIV treatment. AIDS. 2022: 2022. https://programme.aids2022.org/Abstract/Abstract/?abstractid=6805 (accessed October 21, 2024).
[68] Koss A, Ross J. Gilead’s Twice-Yearly Lenacapavir for HIV Prevention Reduced HIV Infections by 96% and Demonstrated Superiority to Daily Truvada® in Second Pivotal Phase 3 Trial. GILEAD 2024. https://www.gilead.com/news/news-details/2024/gileads-twiceyearly-lenacapavir-for-hiv-prevention-reduced-hiv-infections-by-96-and-demonstrated-superiority-to-daily-truvada (accessed October 21, 2024).
[69] WHO. Long-acting injectable lenacapavir proves effective in HIV prevention for women. World Health Organization 2024. https://www.who.int/news/item/26-07-2024-long-acting-injectable-lenacapavir-proves-effective-in-hiv-prevention-for-women#:~:text=Long%2Dacting%20injectable%20lenacapavir%20proves%20effective%20in%20HIV%20prevention%20for%20women,-26%20July%202024&text=WHO%20welcomes%20the%20news%20that,significant%20breakthrough%20in%20HIV%20prevention. (accessed October 21, 2024).
[70] Koss A. Gilead Signs Royalty-Free Voluntary Licensing Agreements with Six Generic Manufacturers to Increase Access to Lenacapavir for HIV Prevention in High-Incidence, Resource-Limited Countries. GILEAD 2024. https://www.gilead.com/news/news-details/2024/gilead-signs-royalty-free-voluntary-licensing-agreements-with-six-generic-manufacturers-to-increase-access-to-lenacapavir-for-hiv-prevention-in-high-incidence-resource-limited-countries (accessed October 21, 2024).
[71] Agência Nacional de Vigilância Sanitária - Anvisa. Anvisa aprova novo medicamento para prevenção do HIV. Ministério Da Saúde Do Brasil 2023. https://www.gov.br/anvisa/pt-br/assuntos/noticias-anvisa/2023/anvisa-aprovou-um-novo-medicamento-para-a-profilaxia-do-hiv (accessed October 21, 2024).
[72] Dourado I, Soares F, Magno L, et al. Adherence, Safety, and Feasibility of HIV Pre-Exposure Prophylaxis Among Adolescent Men Who Have Sex With Men and Transgender Women in Brazil (PrEP1519 Study). J Adolesc Health. 2023; 73: S33-S42. https://doi.org/10.1016/j.jadohealth.2023.09.005.
[73] Grinsztejn B, Torres TS, Hoagland B, et al. Long-Acting Injectable Cabotegravir for HIV Preexposure Prophylaxis Among Sexual and Gender Minorities: Protocol for an Implementation Study. JMIR Public Health Surveill. 2023; 9: e44961. https://doi.org/10.2196/44961.
[74] Ministerio de Salud y Protección Social de Colombia. Profilaxis Preexposición al VIH (PrEP) Lineamiento para la implementación de la PrEP en Colombia. PREP-COLOMBIA 2023. https://prep-colombia.org/wp-content/uploads/2023/07/Lineamiento-PrEP_FINAL_Junio2023.pdf (accessed October 21, 2024).
[75] Departamento Programa Nacional de Prevención y Control del VIH/SIDA e ITS. Orientaciones Técnicas 2019. Profilaxis Pre-Exposición (PrEP) a la infección por VIH. Ministerio de Salud Chile 2019. https://www.prepwatch.org/wp-content/uploads/2022/03/Chile-National-PrEP-Guidelines-2019.pdf (accessed October 21, 2024).
[76] Ministério da Saúde do Brasil. Protocolo Clínico e Diretrizes Terapêuticas para Profilaxia Pré-Exposição (PrEP) de Risco à Infecção pelo HIV. Brasília, DF: Ministério da Saúde; 2022.
[77] Dirección de Prevención y Control de VIH-SIDA, Enfermedades de Transmisión Sexual y Hepatitis. Norma Técnica De Salud “Prevención Combinada Del Virus De La Inmunodeficiencia Humana Para Poblaciones En Alto Riesgo.” Ministerio de Salud Del Perú 2023. https://bvs.minsa.gob.pe/local/MINSA/6345.pdf (accessed October 21, 2024).
[78] Centro Nacional para la preveción y el control del VIH y el SIDA. Guía de manejo antirretroviral de las personas con VIH. Secretaría de Salud Mexico 2021. https://www.gob.mx/cms/uploads/attachment/file/712164/Gu_a_TAR_fe_erratas_2022.pdf (accessed October 21, 2024).
[79] Dirección General de Servicios de Salud. Triptico de Oferta PreP 08. Ministerio de Salud Nicaragua 2022. https://www.minsa.gob.ni/publicaciones/direccion-general-de-servicios-de-salud/triptico-de-oferta-prep-08 (accessed October 21, 2024).
[80] Ministerio de Salud Pública Uruguay. Guía de recomendaciones de tratamiento antirretroviral. Ministerio de Salud Pública Uruguay 2023. https://www.gub.uy/ministerio-salud-publica/comunicacion/publicaciones/guia-recomendaciones-tratamiento-antirretroviral (accessed October 21, 2024).
[81] Ministerio de Salud Argentina. Guía de Uso de PrEP como Parte de la Estrategia de Prevención Combinada. Ministerio de Salud Argentina 2022. http://bancos.salud.gob.ar/sites/default/files/2022-08/Guia_Uso_de_PrEP_como_parte_estrategia_prevencion_combinada.pdf (accessed October 21, 2024).
[82] Ministerio de Salud El Salvador. Lineamientos técnicos para la Profilaxis Preexposición al VIH (PrEP). Ministerio de Salud El Salvador 2021. https://docs.bvsalud.org/biblioref/2022/01/1352917/lineamientostecnicosparalaprofilaxispreexposicionalvihprep-acu_FjWSfbc.pdf (accessed October 21, 2024).
[83] PAHO. Pre-Exposure Prophylaxis (PrEP). PAHO n.d. https://www.paho.org/en/topics/pre-exposure-prophylaxis-prep (accessed October 21, 2024).
[84] Díaz J. PrEP en Venezuela: quiénes pueden utilizarlo y qué aconsejan los especialistas en VIH sobre su uso. El Diario Venezuela 2024. https://eldiario.com/2024/02/29/prep-venezuela-uso-vih/ (accessed October 21, 2024).
[85] Colectivo Amigos Contra el Sida Guatemala. Memoria de labores. CAS Guatemala. Colectivo Amigos Contra El Sida Guatemala n.d. https://casgt.org/documentos/memoria-de-labores/ (accessed October 21, 2024).
[86] Dirección General de Servicios de Salud. Programa Nacional ITS/VIH/SIDA. Guía de Terapia Antirretroviral en Adultos. Ministerio de Salud Bolivia 2017. https://www.minsalud.gob.bo/component/jdownloads/?task=download.send&id=778:guia-de-terapia-antirretroviral-en-adolescentes-y-adultos&catid=42&Itemid=646 (accessed October 21, 2024).
[87] Vega-Ramirez H, Torres TS, Guillen-Diaz C, et al. Awareness, knowledge, and attitudes related to HIV pre-exposure prophylaxis and other prevention strategies among physicians from Brazil and Mexico: cross-sectional web-based survey. BMC Health Serv Res. 2022; 22: 532. https://doi.org/10.1186/s12913-022-07900-y.
[88] Assaf RD, Konda KA, Torres TS, et al. Are men who have sex with men at higher risk for HIV in Latin America more aware of PrEP? PLoS ONE. 2021;16:e0255557. https://doi.org/10.1371/journal.pone.0255557.
[89] CNN Chile. PrEP: Qué es y cómo funciona la pastilla que previene el VIH y el Minsal repartirá en 2019. CNN Chile 2018. https://www.cnnchile.com/pais/prep-que-es-y-como-funciona-la-pastilla-que-previene-el-vih-y-el-minsal-repartira-en-2019_20181228/ (accessed October 21, 2024).
[90] Consejo Nacional de Fijación y Revisión de Precios de Medicamentos de Uso y Consumo Humano. SECRETARÍA TÉCNICA DE FIJACIÓN DE PRECIOS DE MEDICAMENTOS. Ministerio de Salud Pública Ecuador 2020. https://www.salud.gob.ec/consejo-nacional-de-fijacion-y-revision-de-precios-de-medicamentos/ (accessed October 21, 2024).
[91] Luz PM, Osher B, Grinsztejn B, et al. The cost-effectiveness of HIV pre-exposure prophylaxis in men who have sex with men and transgender women at high risk of HIV infection in Brazil. J Intern AIDS Soc. 2018; 21: e25096. https://doi.org/10.1002/jia2.25096.
[92] POZ. Brazil Offers Free PrEP to Prevent HIV Among Youth. POZ 2017. https://www.poz.com/article/brazil-offers-free-prep-prevent-hiv-among-youth (accessed October 21, 2024).
[93] PAHO. PAHO Strategic Fund Long Term Agreements Price List (updated 10/07/2022). PAHO 2022. https://www.paho.org/en/documents/paho-strategic-fund-long-term-agreements-price-list-updated-10072022 (accessed October 21, 2024).
[94] Agemed bolivia. Listado oficial diario de registros sanitarios aprobados. Agemed Bolivia 2024. https://www.agemed.gob.bo/#autorizacioncomercializacion/contenido#registro_sanitario (accessed October 21, 2024).
[95] AIDS Vaccine Advocacy Coalition. HIV prevention is a pathway to global health equity. AVAC n.d. https://avac.org/ (accessed October 21, 2024).
[96] AIDS Vaccine Advocacy Coalition. Country Data. PrEP Watch 2024. https://www.prepwatch.org/data-by-country/ (accessed October 21, 2024).
[97] Ministerio de Salud Pública y Asistencia Social. Estrategia Nacional Para La Implementación de La Profilaxis Pre-Exposición (PrEP) En La República Dominicana. Ministerio de Salud Pública y Asistencia Social República Dominicana 2020. https://repositorio.msp.gob.do/bitstream/handle/123456789/2084/9789945621310.pdf?sequence=1&isAllowed=y (accessed October 21, 2024).
[98] Kadiamada-Ibarra H, Hawley NL, Sosa-Rubí SG, Wilson-Barthes M, Franco RR, Galárraga O. Barriers and facilitators to pre-exposure prophylaxis uptake among male sex workers in Mexico: an application of the RE-AIM framework. BMC Public Health. 2021; 21: 2174. https://doi.org/10.1186/s12889-021-12167-9.
[99] PrEPWatch. Contry data. PrEPWatch n.d. https://www.prepwatch.org/data-by-country/ (accessed January 25, 2025).
[100] Antonini M, Silva IED, Elias HC, Gerin L, Oliveira AC, Reis RK. Barriers to Pre-Exposure Prophylaxis (PrEP) use for HIV: an integrative review. Rev Bras Enferm. 2023; 76: e20210963. https://doi.org/10.1590/0034-7167-2021-0963.
[101] AIDSVu. Prevention & Testing in the United States. AIDSVu 2022. https://map.aidsvu.org/profiles/nation/usa/prevention-and-testing#1-1-PrEP (accessed January 25, 2025).
[102] UK Health Security Agency. Landmark study confirms effectiveness of PrEP. GOVUK 2023. https://www.gov.uk/government/news/landmark-study-confirms-effectiveness-of-prep (accessed January 25, 2025).
[103] Mathews C, Lombard C, Puren A, et al. Evaluation of a South African Combination HIV Prevention Programme for Adolescent Girls and Young Women: HERStory Study. Harvard University 2020. https://scholar.harvard.edu/sonnypatel/publications/evaluation-south-african-combination-hiv-prevention-programme-adolescent (accessed January 25, 2025).
[104] Medland NA, McManus H, Bavinton BR, et al. HIV incidence in people receiving government-subsidised pre-exposure prophylaxis in Australia: a whole-of-population retrospective cohort study. The Lancet HIV. 2024; 11: e756-e764. https://doi.org/10.1016/S2352-3018(24)00213-3.
[105] Brooks RA, Nieto O, Landrian A, Donohoe TJ. Persistent stigmatizing and negative perceptions of pre-exposure prophylaxis (PrEP) users: implications for PrEP adoption among Latino men who have sex with men. AIDS Care. 2019; 31: 427-435. https://doi.org/10.1080/09540121.2018.1499864.
[106] Eaton LA, Kalichman SC, Price D, Finneran S, Allen A, Maksut J. Stigma and Conspiracy Beliefs Related to Pre-exposure Prophylaxis (PrEP) and Interest in Using PrEP Among Black and White Men and Transgender Women Who Have Sex with Men. AIDS Behav. 2017; 21: 1236-1246. https://doi.org/10.1007/s10461-017-1690-0.
[107] Haire B. Preexposure prophylaxis-related stigma: strategies to improve uptake and adherence – a narrative review. HIV. 2015: 241. https://doi.org/10.2147/HIV.S72419.
[108] Luz PM, Veloso VG, Grinsztejn B. The HIV epidemic in Latin America: accomplishments and challenges on treatment and prevention. Current Opinion in HIV and AIDS 2019; 14: 366-373. https://doi.org/10.1097/COH.0000000000000564.
[109] Maroni A. Profilaxis Pre-Exposición en América Latina (Argentina, Brasil y México). Estud Sociol. 2022; 40: 895-912. https://doi.org/10.24201/es.2022v40n120.2239.
[110] Bozzani FM, Terris-Prestholt F, Quaife M, Gafos M, Indravudh PP, Giddings R, et al. Costs and Cost-Effectiveness of Biomedical, Non-Surgical HIV Prevention Interventions: A Systematic Literature Review. PharmacoEconomics. 2023; 41: 467-480. https://doi.org/10.1007/s40273-022-01223-w.
[111] Bórquez A, Guanira JV, Revill P, Caballero P, Silva-Santisteban A, Kelly S, et al. The impact and cost-effectiveness of combined HIV prevention scenarios among transgender women sex-workers in Lima, Peru: a mathematical modelling study. The Lancet Public Health. 2019; 4: e127-e136. https://doi.org/10.1016/S2468-2667(18)30236-6.
[112] Gomez GB, Borquez A, Case KK, Wheelock A, Vassall A, Hankins C. The Cost and Impact of Scaling Up Pre-exposure Prophylaxis for HIV Prevention: A Systematic Review of Cost-Effectiveness Modelling Studies. PLoS Med. 2013; 10: e1001401. https://doi.org/10.1371/journal.pmed.1001401.
[113] Ravasi G, Grinsztejn B, Baruch R, et al. Towards a fair consideration of PrEP as part of combination HIV prevention in Latin America. J Int AIDS Society. 2016; 19: 21113. https://doi.org/10.7448/IAS.19.7.21113.
[114] Kojima N, Davey DJ, Klausner JD. Pre-exposure prophylaxis for HIV infection and new sexually transmitted infections among men who have sex with men. AIDS. 2016; 30: 2251-2252. https://doi.org/10.1097/QAD.0000000000001185.
[115] Traeger MW, Cornelisse VJ, Asselin J, et al. Association of HIV Preexposure Prophylaxis With Incidence of Sexually Transmitted Infections Among Individuals at High Risk of HIV Infection. JAMA. 2019; 321: 1380-1390. https://doi.org/10.1001/jama.2019.2947.
[116] WHO. WHO implementation tool for pre-exposure prophylaxis (PrEP) of HIV infection. World Health Organization 2024. https://iris.who.int/bitstream/handle/10665/378164/9789240097230-eng.pdf?sequence=1&isAllowed=y (accessed January 25, 2025).
[117] Werner RN, Gaskins M, Nast A, Dressler C. Incidence of sexually transmitted infections in men who have sex with men and who are at substantial risk of HIV infection - A meta-analysis of data from trials and observational studies of HIV pre-exposure prophylaxis. PLoS One. 2018; 13: e0208107. https://doi.org/10.1371/journal.pone.0208107.
[118] Cerecero-García D, Terris-Prestholt F, Macías-González F, Bautista-Arredondo S. State of HIV costing in Latin America and the Caribbean: a systematic literature review. Rev Panam Salud Pública. 2024; 48: 1. https://doi.org/10.26633/RPSP.2024.84.
[119] Krakower DS, Mayer KH. Pre-Exposure Prophylaxis to Prevent HIV Infection: Current Status, Future Opportunities and Challenges. Drugs. 2015; 75: 243-251. https://doi.org/10.1007/s40265-015-0355-4.
[120] ImPrEP. Entendiendo las barreras y los desafíos para la adhesión a la prep: experiencias de discontinuidad de la profilaxis en hsh y mujeres trans en el perú. ImPrEP 2021. https://imprep.org/peru/entendiendo-las-barreras-y-los-desafios-para-la-adhesion-a-la-prep-experiencias-de-discontinuidad-de-la-profilaxis-en-hsh-y-mujeres-trans-en-el-peru/ (accessed January 25, 2025).
[121] Mgodi NM, Murewanhema G, Moyo E, Samba C, Musuka G, Dzinamarira T, et al. Advancing the use of Long-Acting Extended Delivery formulations for HIV prevention in sub-Saharan Africa: challenges, opportunities, and recommendations. J Int AIDS Soc. 2023; 26: e26115. https://doi.org/10.1002/jia2.26115.
[122] Jongen VW, Hoornenborg E, Van Den Elshout MA, Boyd A, Zimmermann HM, Coyer L, et al. Adherence to event-driven HIV PrEP among men who have sex with men in Amsterdam, the Netherlands: analysis based on online diary data, 3-monthly questionnaires and intracellular TFV-DP. J Int AIDS Soc. 2021; 24: e25708. https://doi.org/10.1002/jia2.25708.
[123] Sánchez-Mendoza V, Soriano-Ayala E, Vallejo-Medina P, Martinez O. A Scoping Review of Biobehavioural Interventions to Improve PrEP Continuum of Care in Latin America. Acta Colomb Psicol. 2023; 26: 141-154. https://doi.org/10.14718/ACP.2023.26.2.12.
[124] Hoagland B, Moreira RI, De Boni RB, Kallas EG, Madruga JV, Vasconcelos R, et al. High pre-exposure prophylaxis uptake and early adherence among men who have sex with men and transgender women at risk for HIV Infection: the PrEP Brasil demonstration project. J Int AIDS Society. 2017; 20: 21472. https://doi.org/10.7448/IAS.20.1.21472.
[125] Lalla-Edward ST, Venter WDF. Feasibility and Impact of Community Pharmacy and Novel Pick-up Points for Antiretroviral Therapy Pre-exposure Prophylaxis Initiation and Continuation in Low and Middle-income Countries. Curr HIV/AIDS Rep. 2025; 22: 2. https://doi.org/10.1007/s11904-024-00710-3.
[126] Grangeiro A, Santos LAD, Estevam DL, et al. Telehealth effectiveness for pre-exposure prophylaxis delivery in Brazilian public services: the Combine! Study. J Int AIDS Soc. 2023; 26: e26173. https://doi.org/10.1002/jia2.26173.
[127] Vanhamel J, Rotsaert A, Reyniers T, et al. The current landscape of pre-exposure prophylaxis service delivery models for HIV prevention: a scoping review. BMC Health Serv Res. 2020; 20: 704. https://doi.org/10.1186/s12913-020-05568-w.
[128] Martin CE, Blaauw D, Nongena P, Chidumwa G, Dada S, Jack S, et al. Preferences for Delivery of HIV Prevention Services Among Healthcare Users in South Africa: A Discrete Choice Experiment. AIDS Behav. 2025; 29: 331-341. https://doi.org/10.1007/s10461-024-04519-4.
[129] WHO. WHO continues to support its conditional recommendation for the dapivirine vaginal ring as an additional prevention option for women at substantial risk of HIV. World Health Organization 2021. https://www.who.int/news/item/09-12-2021-who-continues-to-support-its-conditional-recommendation-for-the-dapivirine-vaginal-ring (accessed October 22, 2024).
[130] European, AIDS Clinical Society. EACS Guidelines. EACS 2023. https://www.eacsociety.org/media/guidelines-12.0.pdf (accessed October 22, 2024).
[131] ClinicalTrials.gov. Study of Lenacapavir for HIV Pre-Exposure Prophylaxis in People Who Are at Risk for HIV Infection (PURPOSE 2). ClinicalTrialsGov 2024. https://clinicaltrials.gov/study/NCT04925752 (accessed October 22, 2024).
[132] Krakower D, Mayer KH. HIV pre-exposure prophylaxis. UpToDate 2024. https://www.uptodate.com/contents/hiv-pre-exposure-prophylaxis?search=prep+hiv+treatment&source=search_result&selectedTitle=1%7E150&usage_type=default&display_rank=1 (accessed October 22, 2024).
[133] Brown ER, Hendrix CW, Van Der Straten A, et al. Greater dapivirine release from the dapivirine vaginal ring is correlated with lower risk of HIV-1 acquisition: a secondary analysis from a randomized, placebo-controlled trial. J Int AIDS Soc. 2020;23:e25634. https://doi.org/10.1002/jia2.25634.
[134] Mayo AJ, Browne EN, Montgomery ET, Torjesen K, Palanee-Phillips T, Jeenarain N, et al. Acceptability of the Dapivirine Vaginal Ring for HIV-1 Prevention and Association with Adherence in a Phase III Trial. AIDS Behav. 2021; 25: 2430-2440. https://doi.org/10.1007/s10461-021-03205-z.
[135] Ministerio de Salud Costa Rica. Lineamiento para la profilaxis pre-exposicion al VIH en Costa Rica. CONASIDA 2021. http://conasida.go.cr/docs/Lineamiento-Profilaxis-Pre-Exposicio%CC%81n-al-VIH-MS-Costa-Rica.pdf (accessed October 22, 2024).
[136] Betancourt Llody YA. La infección por el VIH: abordaje de su prevención en hombres que tienen relaciones sexuales con hombres en Cuba. Rev Médica Electrónica n.d.; 43: 3167-3178.
[137] Ministerio de Salud Pública Ecuador. Protocolo Profilaxis previa a la exposición frente a la infección por VIH en Ecuador 2023. Ministerio de Salud Pública Ecuador 2023. https://www.salud.gob.ec/wp-content/uploads/2024/04/Protocolo_para_la_PrEP_2023.pdf (accessed October 22, 2024).
[138] Secretaría de Salud Honduras. Lineamientos para proveer la profilaxis previa a la exposición al virus de la inmunodeficiencia humana. Secretaría de Salud Honduras 2022. https://salud.gob.hn/sshome/index.php/component/jdownloads/?task=download.send&id=1105 (accessed October 22, 2024).
[139] Ministerio de Salud Panamá. Guía para el Abordaje de la Profilaxis Post-Exposición a Infecciones de Transmisión Sexual y al Virus de la Inmunodeficiencia Humana (VIH) de tipo ocupacional y no ocupacional. Ministerio de Salud Panamá 2023. https://www.minsa.gob.pa/sites/default/files/normatividad/resolucion_no_953_de_27_de_diciembre_de_2023_-_guia_abordaje_de_profilaxis_ets_y_vih_ocupacional_y_no_ocupacional_-_normativa.pdf (accessed October 22, 2024).
[140] Programa Nacional de Control del SIDA/ITS. PROTOCOLO CLÍNICO y DIRECTRICES TERAPÉUTICAS PARA PROFILAXIS PRE EXPOSICIÓN. Ministerio de Salud Pública y Bienestar Social Paraguay 2020. https://www.mspbs.gov.py/pronasida/nmanuales.html (accessed October 22, 2024).